
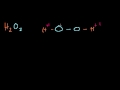
Understanding Oxidation States in Hydrogen Peroxide
Interactive Video
•
Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Hard
Emma Peterson
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What was the main error pointed out in the second oxidation video?
The structure of hydrogen peroxide was incorrect.
The oxidation state of oxygen in hydrogen peroxide was incorrectly stated.
The number of hydrogen atoms in hydrogen peroxide was wrong.
The electronegativity of oxygen was not mentioned.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How are the atoms in hydrogen peroxide bonded?
The hydrogens are bonded to each other and each to an oxygen.
The oxygens are bonded to each other and each to a hydrogen.
Each hydrogen is bonded to two oxygens.
Each oxygen is bonded to two hydrogens.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why does oxygen have a tendency to 'hog' electrons?
Because it has a high atomic number.
Because it is highly electronegative.
Because it has a low electronegativity.
Because it is a noble gas.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the oxidation state of hydrogen in hydrogen peroxide?
-1
+1
0
+2
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does gaining an electron affect the oxidation state?
It increases the oxidation state.
It makes the oxidation state positive.
It has no effect on the oxidation state.
It decreases the oxidation state.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What mistake did the narrator make regarding the oxidation state of oxygen?
Stated it as 0 instead of -1.
Stated it as -2 instead of -1.
Stated it as +1 instead of -1.
Stated it as +2 instead of -1.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the correct oxidation state of oxygen in hydrogen peroxide?
0
-1
-2
+1
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
