
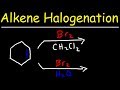
Halogenation and Stereochemistry of Alkenes
Interactive Video
•
Chemistry
•
10th Grade - University
•
Practice Problem
•
Hard
Sophia Harris
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the role of the double bond in the halogenation of alkenes?
It acts as an electrophile.
It acts as a nucleophile.
It acts as a catalyst.
It acts as a solvent.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of addition occurs in the halogenation of alkenes?
No addition
Random addition
Anti-addition
Syn-addition
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What products are formed when cis-2-butene reacts with bromine?
A single meso compound
A pair of enantiomers
A single racemic mixture
No reaction occurs
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does the symmetry of trans-2-butene affect the reaction with bromine?
It forms a meso compound.
It forms a pair of enantiomers.
It forms a single product.
It forms no products.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why does trans-2-pentine not form meso compounds?
It is an unsymmetrical alkene.
It forms only syn-addition products.
It is a symmetrical alkene.
It does not react with bromine.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In halohydrin formation, what is the role of water?
It acts as a nucleophile.
It acts as an electrophile.
It acts as a catalyst.
It acts as a solvent.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which carbon does the OH group attach to in halohydrin formation?
The less substituted carbon
The more substituted carbon
The carbon with no substituents
The carbon with a negative charge
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
