
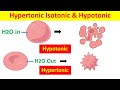
Cell Tonicity: Exploring Hypertonic, Isotonic, and Hypotonic Solutions
Interactive Video
•
Biology, Science, Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the prefix 'hyper' signify in the context of solutions?
Less
Same
More
Equal
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is tonicity defined in relation to solutions?
As a measure of temperature
As a relative measure compared to another solution
As an absolute measure of solute concentration
As a measure of pH level
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to a cell when placed in a hypotonic solution?
The cell shrinks
The cell remains unchanged
The cell swells and may burst
The cell becomes rigid
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a hypotonic solution, where does the water move?
There is no movement
It moves in both directions equally
From the solution to the cell
From the cell to the solution
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What characterizes an isotonic solution?
No solute present
Equal solute concentration as the cell
Lower solute concentration than the cell
Higher solute concentration than the cell
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the net movement of water in an isotonic solution?
Both into and out of the cell
Out of the cell
Into the cell
No net movement
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What defines a hypertonic solution?
It has no solute
It has more solute than the cell
It has the same solute concentration as the cell
It has less solute than the cell
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
