
Gas Laws and Mole Fractions
Interactive Video
•
Chemistry, Physics, Science
•
9th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does Dalton's Law of Partial Pressure state?
The total pressure of a gas mixture is the sum of the partial pressures of its components.
The total pressure of a gas mixture is the difference between the partial pressures of its components.
The total pressure of a gas mixture is the average of the partial pressures of its components.
The total pressure of a gas mixture is the product of the partial pressures of its components.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the example of oxygen collected over water, what is the vapor pressure of water at 24 degrees Celsius?
745 Torr
24.38 Torr
721 Torr
50 Atmospheres
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you calculate the partial pressure of a gas using Dalton's Law?
By dividing the total pressure by the vapor pressure of water.
By adding the total pressure to the vapor pressure of water.
By subtracting the vapor pressure of water from the total pressure.
By multiplying the total pressure by the vapor pressure of water.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
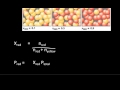
What is the mole fraction of a gas in a mixture?
The ratio of the gas's mass to the total mass of the mixture.
The ratio of the gas's volume to the total volume of the mixture.
The ratio of the gas's moles to the total moles of the mixture.
The ratio of the gas's pressure to the total pressure of the mixture.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the example with neon and oxygen, what is the total pressure in the gas tanks?
50 Atmospheres
721 Torr
24.38 Torr
745 Torr
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you convert mass to moles for calculating mole fraction?
By dividing the mass by the molar mass.
By multiplying the mass by the molar mass.
By adding the mass to the molar mass.
By subtracting the mass from the molar mass.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the mole fraction of oxygen in the mixture of neon and oxygen?
0.20999
0.5
1.0
0.9
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
