
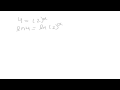
Determining Reaction Orders and Rate Laws
Interactive Video
•
Chemistry, Science, Mathematics
•
11th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary purpose of the method of initial rates?
To determine the rate law from experimental data
To calculate the final concentrations of reactants
To find the equilibrium constant of a reaction
To determine the rate law from a balanced equation
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the experimental setup, what information is recorded for each trial?
Temperature and pressure
Volume and mass of reactants
Time and color change
Concentration of reactants and reaction rate
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
When comparing trials to determine the order of reactant A, what condition must be met?
The concentration of reactant A must be the same
The temperature must be constant
The concentration of reactant B must be the same
The reaction rate must be identical
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the order of reactant A determined using the method of initial rates?
By comparing the rates of trials with the same concentration of B
By measuring the temperature change
By using the balanced chemical equation
By comparing the rates of trials with different concentrations of A
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What mathematical operation is used to solve for the order of reactant A?
Natural logarithm
Subtraction
Addition
Multiplication
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the overall order of the reaction once both reactant orders are determined?
Zero order
Third order
Second order
First order
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the order of reactant B determined?
By comparing trials with different concentrations of B
By using the same method as for reactant A
By measuring the pressure change
By using the initial concentrations only
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
