
Energy Levels and Light Emission
Interactive Video
•
Physics, Chemistry, Science
•
9th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the purpose of using gas spectra in atomic studies?
To identify the chemical composition of gases
To calculate the mass of atoms
To understand the internal structure of atoms
To measure the temperature of gases
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens when a gas is subjected to an electric discharge or heat?
It becomes solid
It changes color
It emits light
It absorbs light
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
According to Bohr, why don't electrons emit light continuously?
They are not charged
They are in specific orbits where they don't lose energy
They are stationary
They are too far from the nucleus
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
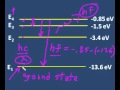
What is the ground state energy level for hydrogen according to Bohr?
-3.4 electron volts
13.6 electron volts
-13.6 electron volts
0 electron volts
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens when an electron moves from a higher energy level to a lower one?
It becomes neutral
It gains energy
It emits a photon
It absorbs a photon
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the energy difference between two levels related to the wavelength of light emitted?
Inversely proportional
Not related
Directly proportional
Equal to the wavelength
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the energy level of E2 in hydrogen?
-1.5 electron volts
0 electron volts
-3.4 electron volts
-13.6 electron volts
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
