
Propane Reaction and Gas Volumes
Interactive Video
•
Chemistry, Physics, Science
•
9th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the initial volume of propane gas used in the reaction?
4 L
1 L
2 L
3 L
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
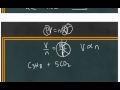
What is the balanced chemical equation for the reaction involving propane and oxygen?
C3H8 + 5O2 → 3CO2 + 4H2O
C3H8 + 5O2 → 2CO2 + 4H2O
C3H8 + 4O2 → 3CO2 + 4H2O
C3H8 + 6O2 → 3CO2 + 4H2O
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it important to identify the limiting reagent in a chemical reaction?
To determine the amount of excess reactant
To calculate the total pressure
To balance the chemical equation
To find the maximum amount of product formed
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the volume of gases related to the number of moles under constant conditions?
Volume is directly proportional to moles
Volume is unrelated to moles
Volume is equal to moles
Volume is inversely proportional to moles
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the limiting reagent in the reaction between propane and oxygen?
Carbon Dioxide
Propane
Oxygen
Water
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many liters of carbon dioxide are formed from 6.3 L of oxygen?
5.78 L
2.78 L
3.78 L
4.78 L
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the total volume of gas in the reaction vessel at the end of the reaction?
3.52 L
5.52 L
4.52 L
6.52 L
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
