
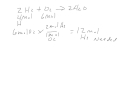
Limiting Reactants in Chemical Reactions
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the role of a limiting reactant in a chemical reaction?
It is always present in excess.
It determines the amount of product formed.
It has no effect on the reaction.
It speeds up the reaction.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the grilled cheese sandwich analogy, what was the limiting reactant?
Cheese
Butter
Tomato
Bread
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in solving a limiting reactant problem?
Determine the excess reactant.
Calculate the reaction rate.
Identify the products.
Write a balanced chemical equation.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the reaction 2 H2 + O2 → 2 H2O, which reactant is limiting if you have 4 moles of H2 and 6 moles of O2?
None
O2
H2
H2O
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many moles of H2O are formed from 4 moles of H2 in the reaction with O2?
4 moles
6 moles
8 moles
2 moles
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a mass-mass problem, what is the first step to determine the limiting reactant?
Calculate the molar mass of the reactants.
Write the balanced chemical equation.
Determine the volume of the reactants.
Identify the products.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If 25 g of iron oxide can produce 19.4 g of iron, what is the limiting reactant?
Iron oxide
Aluminum
Oxygen
Iron
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
