
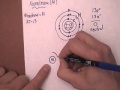
Bohr Model and Atomic Structure
Interactive Video
•
Chemistry
•
10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary purpose of the Bohr Model?
To determine chemical reactions
To measure atomic energy
To represent atomic structure
To calculate atomic mass
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why are elements on the periodic table considered neutral?
They have more protons than electrons
They have equal numbers of protons and electrons
They have no neutrons
They have more electrons than protons
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many protons does aluminum have?
13
8
27
14
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the maximum number of electrons in the L orbit of the Bohr model?
8
2
18
32
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many electrons are in the outermost shell of aluminum in the Bohr model?
5
8
3
2
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is an alternative way to represent the Bohr model?
Using chemical equations
Using partial orbits
Using complete circles
Using atomic numbers
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does a Lewis Dot Diagram emphasize?
The number of protons
The outermost shell
The atomic mass
The atomic number
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
