
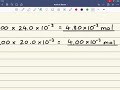
Calculating pH in Acid-Base Reactions
Interactive Video
•
Chemistry
•
10th - 11th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in calculating the pH after mixing strong acids and bases?
Mix the solutions thoroughly.
Find the pH directly from the concentration.
Calculate the moles of hydrogen ions.
Determine the total volume of the solution.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you calculate the moles of hydrogen ions from hydrochloric acid?
By multiplying its concentration by its volume.
By adding the concentration and volume.
By using the pH formula directly.
By dividing the concentration by the volume.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the relationship between the concentration of sodium hydroxide and hydroxide ions?
They are unrelated.
Concentration of hydroxide ions is half.
Concentration of sodium hydroxide is double.
They are equal.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What do you do after calculating the moles of hydrogen and hydroxide ions?
Determine which ion is in excess.
Calculate the pH directly.
Add more acid to the solution.
Mix the solutions again.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you find the concentration of excess hydrogen ions?
By dividing the moles of hydroxide ions by the total volume.
By multiplying the moles of hydrogen ions by the total volume.
By adding the moles of hydroxide ions to hydrogen ions.
By subtracting the moles of hydroxide ions from hydrogen ions and dividing by total volume.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the final step in calculating the pH of the solution?
Subtract the concentration from the volume.
Multiply the concentration by the volume.
Use the formula pH = -log[H+].
Add the concentrations of all ions.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the pH of a solution with a hydrogen ion concentration of 0.0182 M?
1.00
1.74
2.00
3.00
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
