
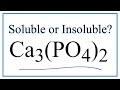
Solubility of Calcium Phosphate
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Jackson Turner
FREE Resource
Read more
8 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of compound is calcium phosphate?
Molecular
Covalent
Ionic
Metallic
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
According to solubility rules, which group of elements generally forms soluble phosphates?
Noble gases
Group 1 elements
Transition metals
Group 2 elements
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is calcium phosphate considered insoluble in water based on solubility rules?
It is a covalent compound
It reacts with water to form a new compound
It forms a gas when dissolved
It does not fall under the exceptions for soluble phosphates
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the solubility chart indicate about calcium phosphate?
It is highly soluble
It is slightly soluble
It forms a precipitate
It is insoluble
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does the solubility chart confirm the solubility rules for calcium phosphate?
It does not list calcium phosphate
It shows calcium phosphate as a gas
It shows calcium phosphate as insoluble
It shows calcium phosphate as soluble
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to calcium phosphate when placed in water?
It reacts violently
It dissolves completely
It remains mostly solid
It forms a gas
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is noted about the small amount of calcium phosphate that might dissolve in water?
It changes the color of the solution
It forms a new compound
It is negligible and can be ignored
It is significant and should be considered
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
