
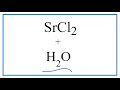
Dissolution of Strontium Chloride Concepts
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Emma Peterson
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the initial state of strontium chloride (SrCl2) when placed in water?
Gas
Solid
Plasma
Liquid
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which rule helps determine if strontium chloride will dissolve in water?
Periodic Table
Molecular Geometry
Electronegativity Chart
Solubility Rules
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge of the strontium ion (Sr) when it dissociates in water?
2+
1-
1+
2-
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many chloride ions are formed when SrCl2 dissolves in water?
One
Two
Four
Three
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the role of water in the dissolution of SrCl2?
It acts as a solvent
It acts as a product
It acts as a reactant
It acts as a catalyst
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which ion is represented as Cl- in the dissolution process?
Chloride ion
Oxygen ion
Hydrogen ion
Strontium ion
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the subscript '2' in SrCl2 indicate?
Two hydrogen ions
Two water molecules
Two strontium ions
Two chloride ions
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
