
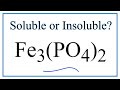
Solubility of Iron II Phosphate
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Ethan Morris
FREE Resource
Read more
8 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the solubility status of Iron II Phosphate in water?
Soluble
Partially soluble
Highly soluble
Insoluble
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
According to solubility rules, which of the following is true about phosphates?
Phosphates dissolve in all conditions
All phosphates are soluble
Most phosphates are insoluble
Phosphates are always exceptions
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is Iron II Phosphate considered insoluble according to solubility rules?
It does not fall under any exceptions
It dissolves completely
It reacts with water
It is an exception to the rules
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the 'I' in the solubility chart indicate for Iron II Phosphate?
Iron II Phosphate is highly reactive
Iron II Phosphate is partially soluble
Iron II Phosphate is insoluble
Iron II Phosphate is soluble
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does Iron II Phosphate behave when placed in water according to the solubility chart?
It floats on the surface
It reacts violently
It sinks to the bottom
It dissolves completely
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the significance of a small amount of Iron II Phosphate dissolving in water?
It indicates a chemical reaction
It is still considered insoluble
It makes it soluble
It changes the water color
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What practical method can be used to test the solubility of Iron II Phosphate?
Heating it
Mixing it with acid
Placing it in water
Exposing it to sunlight
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
