
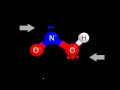
Polarity and Structure of HNO2
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Olivia Brooks
FREE Resource
Read more
8 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in determining whether HNO2 is polar or nonpolar?
Checking the molecular weight
Analyzing the color of the compound
Examining the Lewis structure
Measuring the boiling point
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why does the asymmetry in the Lewis structure of HNO2 suggest it might be polar?
Because asymmetry often leads to nonpolarity
Because asymmetry can create a separation of charge
Because it has a symmetrical distribution of electrons
Because it has a high molecular weight
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does electronegativity contribute to the polarity of HNO2?
It causes the molecule to become nonpolar
It makes the molecule symmetrical
It results in a positive and a negative side
It creates a uniform charge distribution
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which atom in HNO2 is more positive due to electronegativity differences?
Oxygen
All atoms are equally positive
Nitrogen
Hydrogen
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the molecular geometry of HNO2 reveal about its polarity?
It indicates a symmetrical structure
It suggests the molecule is neutral
It confirms the presence of poles
It shows that the molecule is nonpolar
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What color represents oxygen in the molecular model of HNO2?
Blue
White
Green
Red
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the electrostatic potential surface of HNO2 show?
A single uniform charge
Two distinct poles
No charge distribution
A neutral molecule
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
