
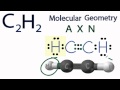
Acetylene Molecular Geometry and Bonding
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Olivia Brooks
FREE Resource
Read more
9 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What are the two types of atoms attached to the carbon atom on the left in acetylene?
Carbon and Oxygen
Hydrogen and Carbon
Hydrogen and Nitrogen
Hydrogen and Oxygen
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molecular geometry of acetylene?
Tetrahedral
Trigonal Planar
Linear
Bent
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do the hydrogen and carbon atoms arrange themselves in acetylene?
In a triangular shape
In a straight line
In a circular pattern
In a zigzag pattern
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of bond is present between the carbon atoms in acetylene?
Triple bond
Quadruple bond
Single bond
Double bond
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In AXN notation, what does 'X' represent for acetylene?
Number of hydrogen atoms
Number of electron pairs
Number of atoms bonded to the central atom
Number of lone pairs
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the value of 'X' in the AXN notation for acetylene?
4
2
1
3
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the bond angle in a linear molecular geometry like acetylene?
360°
180°
120°
90°
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
