

Valence Electrons
Presentation
•
Chemistry
•
9th - 12th Grade
•
Practice Problem
•
Medium
Standards-aligned
Nicholas Carpenter
Used 33+ times
FREE Resource
6 Slides • 12 Questions
1
Valence Electrons
Atoms want it all...
or nothing at all

2
What are valence electrons?
the electrons in the outermost shell
Shell is another way to say energy levels
Atoms want to have a stable outer shell of electrons
3
What makes an outer shell stable?
Being completely full of electrons
or just having full s and p subshells/sublevels (they mean the same thing)
Noble Gases have 8 valence electrons - full s and p subshells
Atoms want to have a "Noble Gas" configuration
4
So how do atoms become stable?
They will either gain electrons and become anions (negatively charged)
or they will give up electrons and become cations (positively charged)
Groups 1-3 (or 1A-3A) give up electrons - gets them to having 8 electrons in their outer shell
Groups 15-17 (or 5A-7A) gain electrons - their outer shell becomes empty, so the shell below it becomes the new outer shell, it is already full
5
How many valence electrons do neutral atoms have?
Old group (groups are columns) number (1A-8A) = number of valence electrons
Lithium- 1 valence electron
Beryllium - 2 valence electrons
skip across to Boron - 3 valence electrons
Carbon - 4; Nitrogen - 5; Oxygen - 6; Fluorine - 7; Neon - 8
6
Determine number of valence electrons with electron configurations
Find the highest shell number (the first number in a term)
(1s2 - shell number is 1; 3p4 - shell number is 3)
Add the number of electrons in that shell
ex. 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p3
highest shell number is 4, so add 4s2 (2 electrons) and 4p3 (3 electrons) = 5 valence electrons
7
Multiple Choice
8
Multiple Choice
What electron configuration matches an oxygen atom?
1s22s22p63s2, 3p64s23d104p5
1s22s22p4
1s22s22p6
1s22s22p63s23p64s23d1
9
Multiple Choice
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice
USE THE PERIODIC TABLE
How many valence electrons does Phosphorus have?
31
5
15
4
13
Multiple Choice
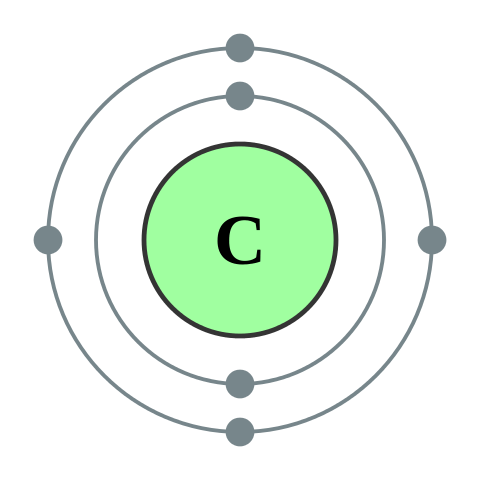
How many valence electrons does carbon have?
4
5
6
7
14
Multiple Choice

15
Multiple Choice

16
Multiple Choice
What are valence electrons?
The total number of electrons in an atom
The number of electrons in the outermost shell
The number of electrons in the second shell
The number of protons in the outermost shell
17
Multiple Choice
How many valence electrons are in the following configuration?
1s2 2s2 2p6 3s2
6
8
2
12
18
Multiple Choice
How many valence electrons are in the following electron configuration?
[Kr] 5s2 4d10 5p2
4
10
2
12
Valence Electrons
Atoms want it all...
or nothing at all

Show answer
Auto Play
Slide 1 / 18
SLIDE
Similar Resources on Wayground

13 questions
Conflict
Presentation
•
9th - 12th Grade

14 questions
Supply and Demand
Presentation
•
9th - 12th Grade

12 questions
Ecological Succession
Presentation
•
9th - 12th Grade

13 questions
Accuracy vs. Precision
Presentation
•
10th - 12th Grade

16 questions
Moles and Percent Composition
Presentation
•
9th - 11th Grade

15 questions
Ions review (forming compounds and naming)
Presentation
•
9th - 12th Grade

13 questions
Hacking
Presentation
•
9th - 12th Grade

13 questions
Ilmu Pengetahuan Alam
Presentation
•
KG
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade