

Phase Changes
Presentation
•
Physics
•
5th - 12th Grade
•
Medium
+2
Standards-aligned
Adam Morris
Used 226+ times
FREE Resource
15 Slides • 8 Questions
1
Phase Changes

2
Multiple Select
What are the 3 states of matter we are most familiar with?
solid
liquid
gas
Texas
3
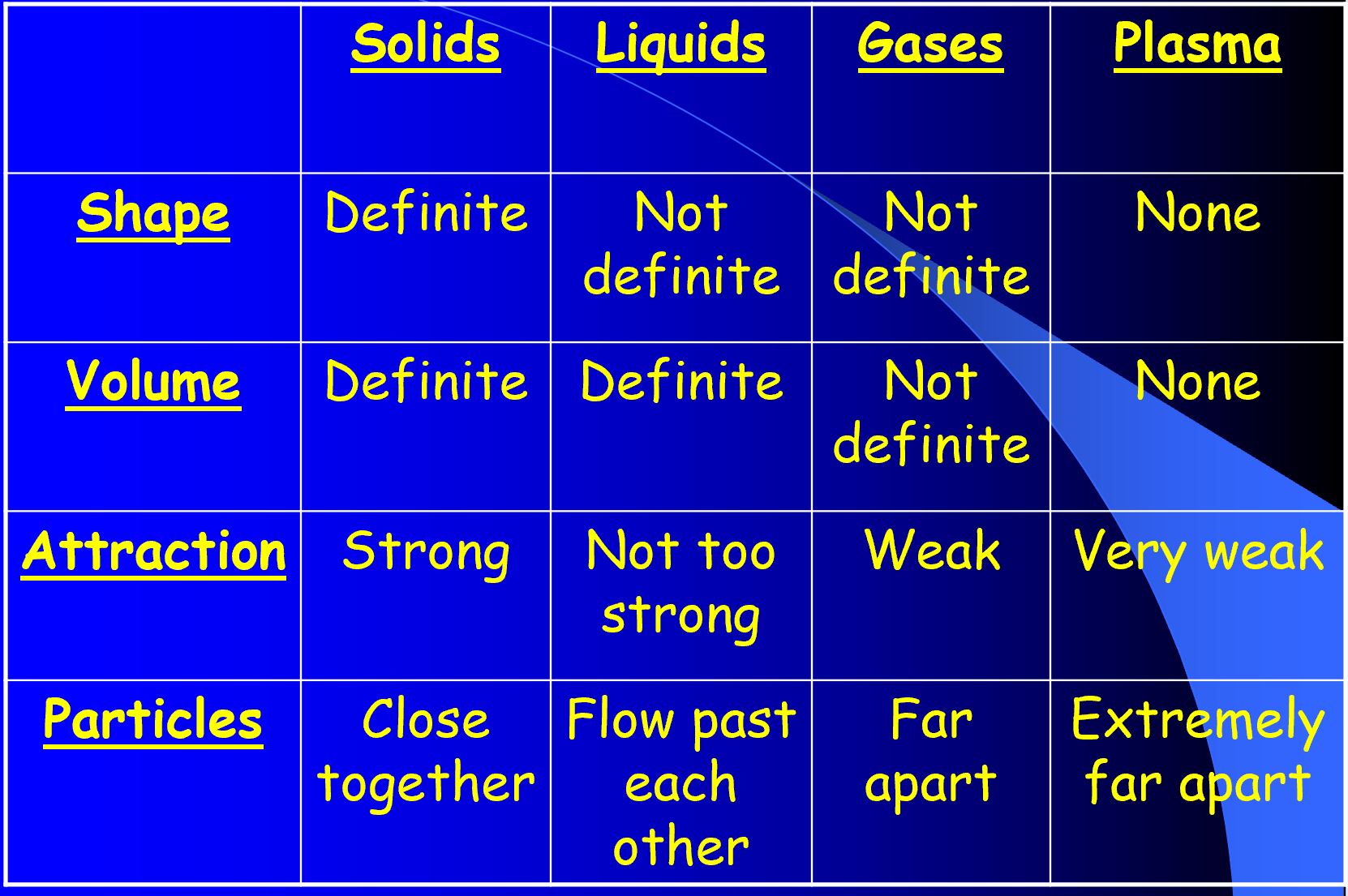
4
The Law of Conservation of Energy states that energy cannot be created nor destroyed, but only changed from one form to another.
5
Multiple Choice
Energy cannot be ___________ or destroyed.
used
created
recycled
6
Thermal energy must be added to change from a solid to a liquid to a gas.
Thermal energy must be removed to change phase from a gas to a liquid to a solid.
7
Poll
Thermal energy probably means ________
heat
electricity
movement
8
Melting – solid changing into a liquid
Vaporization – liquid changing into a gas
Ionization – gas changing into a plasma
Neutralization – plasma changing into a gas
Condensation – gas changing into a liquid
Freezing – liquid changing into a solid
Sublimation – direct change of a solid to a gas or a gas to a solid without going to a liquid state
9
A phase change graph is a temperature vs. time graph displaying the phases of matter and the temperature at which phase changes occur.
10

11
Multiple Choice

On this graph, as time increases, temperature...
increases
decreases
12

13
Multiple Choice

The diagonal lines on a phase change graph represent...
states of matter
phase change from one phase of matter to another
14

15
Multiple Choice
The flat lines on this graph represent
phases of matter
phase changes from one form of matter to another
16

17
As matter changes phase, thermal energy is absorbed or released until all particles of matter have changed. Therefore, the temperature remains constant.
18
Multiple Select
During phase change...
temperature remains the same
the matter is transforming from one form to another
temperature increases
energy is created
19
The melting point is the temperature at which a solid becomes a liquid.
The boiling point is the temperature at which a liquid becomes a gas.
20
The condensation point is the temperature at which a gas becomes a liquid.
Condensation point = Boiling point
The freezing point is the temperature at which a liquid becomes a solid.
Freezing point = Melting point
21
Heating and cooling systems regulate temperature by transferring energy.
A heating system is any device or process that transfers energy to a substance to raise its temperature.
ex. Rubbing hands together, human body, furnace, reptiles basking in the sun, solar panels.
22
A cooling system is any device or process that transfers energy as heat out of a substance to lower its temperature.
ex. Refrigerator, air conditioner, condensation, sweating, heat pumps.
23
Multiple Choice
Heating systems put heat INTO a system and cooling systems...
take heat out
put cold in
Phase Changes

Show answer
Auto Play
Slide 1 / 23
SLIDE
Similar Resources on Wayground

16 questions
Algebra 1 FSA EOC Practice Test ~ Non-Calculator
Presentation
•
10th - 11th Grade

19 questions
Pattern/Repetition
Presentation
•
5th - 12th Grade

18 questions
Subject-Verb Agreement: Compound Subjects, Indefinite Pronouns, Inverted Order
Presentation
•
5th - 12th Grade

20 questions
Present Perfect vs Perfect continuous tense
Presentation
•
5th - 12th Grade

18 questions
O → UE Stem Changers
Presentation
•
6th - 12th Grade

17 questions
Ciencias Sociales
Presentation
•
6th - 12th Grade

17 questions
Biotic and Abiotic Factors
Presentation
•
4th - 12th Grade

20 questions
The Origins of Buddhism
Presentation
•
KG - University
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Physics

11 questions
Electricity Explained
Interactive video
•
9th - 12th Grade

18 questions
Series and Parallel Circuits
Quiz
•
9th Grade

10 questions
Force and Motion
Quiz
•
4th - 5th Grade

15 questions
waves and wave properties
Quiz
•
9th - 12th Grade

35 questions
Electricity and Magnetism SPS10
Quiz
•
8th - 10th Grade

10 questions
Exit Check 7.1 Intro to Waves
Quiz
•
9th Grade

10 questions
Exit Check 7.2: Electromagnetic Waves
Quiz
•
9th Grade

10 questions
Exit Check 7.3 - Analog and Digital
Quiz
•
9th Grade