

Density
Presentation
•
Chemistry
•
10th Grade
•
Easy
+2
Standards-aligned
Laura Zobel
Used 5+ times
FREE Resource
19 Slides • 23 Questions
1
Density

2

3
Fill in the Blanks
Type answer...
4
Multiple Choice
Density is mass divided by
kilograms
grams
volume
5
Multiple Choice
The side of a dice is 2 mm. What is the Volume of the dice?
6 cubic millimeters
8 cubic millimeters
8 cubic meters
8 square millimeters
6
Multiple Choice
What is the Volume of a cube with side lengths of 20 meters?
60 cubic meters
40 cubic meters
8000 cubic meters
20 cubic meters
7
Multiple Choice
Miguel wants to bake a cake in the shape of a cube. The pan is 9 in by 9 in by 9 in. What will be the Volume of Miguel's cake?
729 ft 3
729 in 3
81 in 3
27 in 3
8

9
Multiple Choice
What is the formula for density?
density = mass * volume
density = volume * mass
density = mass/volume
density = volume/mass
10

11
Multiple Choice
Which takes up more space, Kg of feathers or a kg of steel?
feathers
steel
12

13

14
Multiple Choice
Which substances have density?
gold bar
diet coke
carbon dioxide
all of these
15

16
Multiple Choice
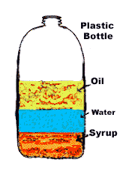
17
Multiple Choice

18
Multiple Choice

19

20

21

22

23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
28

29

30
The object placed in water, displaces water equal to its volume.
Formula:
Final- Initial = volume

31

32
Temperature
warm - high kinetic energy
molecules further apart ( Less dense)
Cold - lower kinetic energy
Molecules are more compact ( more dense)

33
Multiple Select
Temperature has which effect on density?
NO effect
warm less dense
cool more dense
34

35
Concentration
high concentration of substance increases density
low concentration = lighter density

36
Multiple Choice
If the density of a diamond is 3.5 g/cm3 , what would be the mass of a diamond whose volume is .5 cm3?
Hint: density = mass/volume
1.75 g
7 g
0.14 g
4 g
37
Multiple Choice
If a 96.5g piece of aluminum has a density of 2.7 g/cm3, what is its volume?
Hint: density = mass/volume
35.7 cm3
260.55 cm3
0.03 cm3
99.2 cm3
38
Multiple Choice
If 96.5 grams of gold has a volume of 5 cm3 , what is the density of gold?
Hint: density = mass/volume
482.5 g/cm3
19.3 g/cm3
0.05 g/cm3
101.5 g/cm3
39

40
Multiple Choice
41
Multiple Choice

42
Multiple Choice
A piece of tin has a mass of 16.52 g and a volume of 2.26 cm3. What is the density of tin?
Hint: density = mass/volume
7.3 g/cm3
37.3 g/cm3
7.3 cm3/g
37.3 cm3/g
Density

Show answer
Auto Play
Slide 1 / 42
SLIDE
Similar Resources on Wayground

38 questions
Basic Economic Concepts
Presentation
•
9th - 10th Grade

35 questions
Untitled Presentation
Presentation
•
10th Grade

35 questions
Intramolecular Forces
Presentation
•
10th Grade

39 questions
Classification terms 2023
Presentation
•
9th - 10th Grade

36 questions
Mutations
Presentation
•
10th Grade

37 questions
Enlightenment Philosophers
Presentation
•
10th Grade

38 questions
The Crusades
Presentation
•
10th Grade

36 questions
Similarity Unit Test Review
Presentation
•
10th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade