

CSA Fall 1st Qtr
Presentation
•
Chemistry
•
10th - 12th Grade
•
Medium
+1
Standards-aligned
Kari Espada
Used 6+ times
FREE Resource
4 Slides • 33 Questions
1
CSA Fall 1st Qtr
Chemistry

2
3
Multiple Choice
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
What are the 3 subatomic particles of an atom?
carbon atom, proton, electron
quarks, neutrons, protons
nucleus, protons, electrons
protons, neutrons, electrons
7
Multiple Choice
A subatomic particle that has a negative charge
nobles gases
protons
electrons
neutrons
8
Multiple Choice
A subatomic particle that has a positive charge and that is found in the nucleus of an atom
Proton
elecrtron
groups
neutrons
9
Multiple Choice
neutron
A subatomic particle that has a positive charge and that is found in the nucleus of an atom
not able to conduct heat or electricity, little to no metallic luster
A subatomic particle that has a negative charge
A small particle in the nucleus of the atom, with no electrical charge
10
Multiple Choice
11
Multiple Choice
Use your periodic table to find which element has 5 protons.
Lithium
Calcium
Boron
Neon
12
Multiple Choice
The horizontal (side to side) rows in the Periodic Table are called
groups
families
periods
atomic numbers
13
Multiple Choice
14
Multiple Choice
15
Multiple Choice
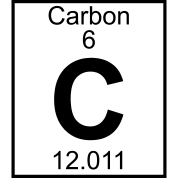
16
Multiple Choice
17
Multiple Choice
18
Multiple Choice
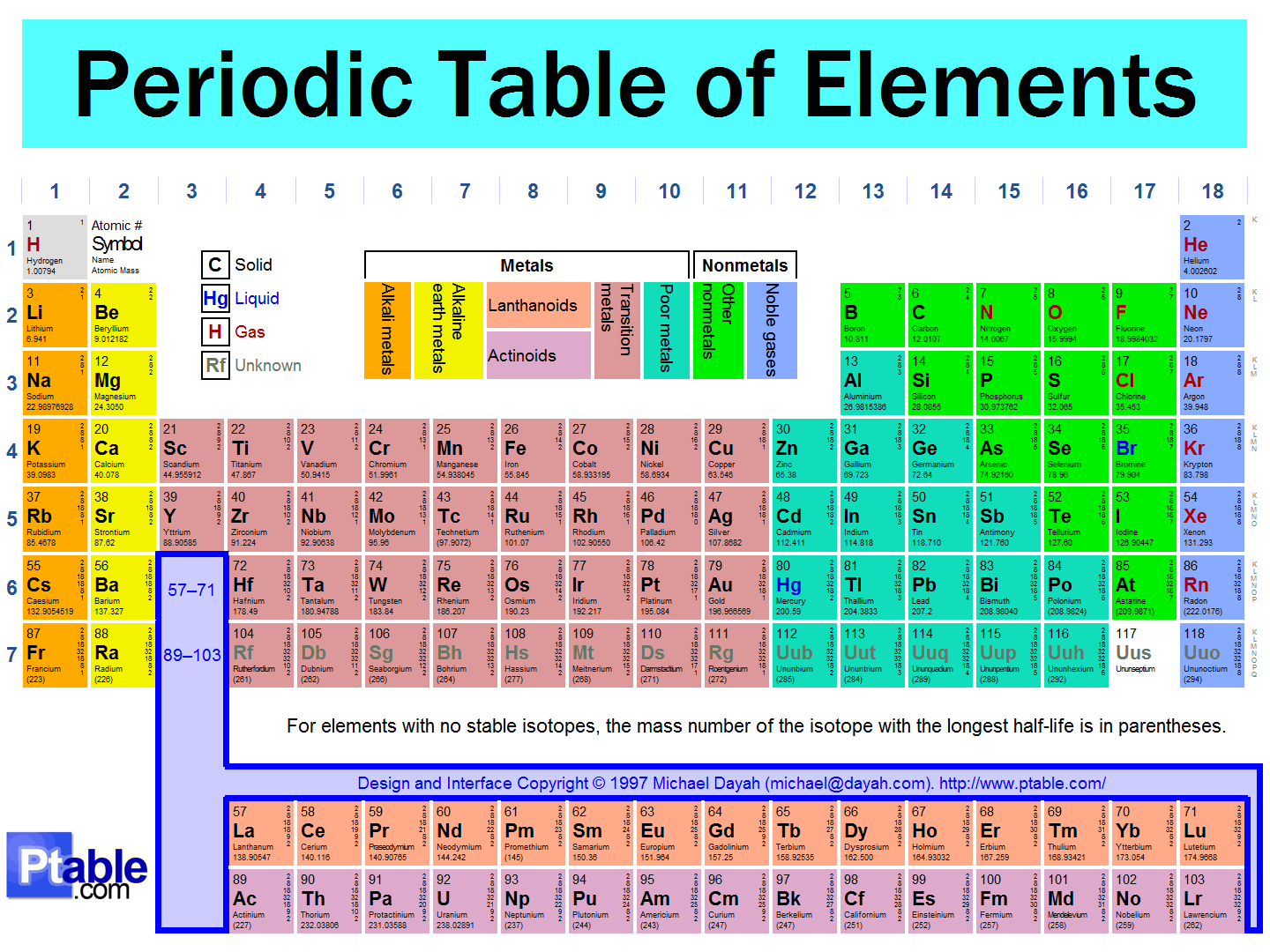
The vertical (up and down) columns in the Periodic Table are called
groups
towers
periods
atomic numbers
19

20
Multiple Choice
21
Multiple Choice
22
Multiple Choice
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
28
Multiple Choice

29
Multiple Choice

30
Multiple Choice

31
Multiple Choice
32
Multiple Choice
33
Multiple Choice
34
Multiple Choice

35
Multiple Choice

36
Multiple Choice
37
Thank You
Go Study

CSA Fall 1st Qtr
Chemistry

Show answer
Auto Play
Slide 1 / 37
SLIDE
Similar Resources on Wayground

32 questions
Significant Figures
Presentation
•
9th - 12th Grade

32 questions
Biomolecules Interactive
Presentation
•
10th - 12th Grade

34 questions
Unit 4 Section 3: Modeling Phase Changes
Presentation
•
10th - 12th Grade

33 questions
Energy & Energy Transformations
Presentation
•
9th - 12th Grade

30 questions
Endothermic/Exothermic & Calorimetry
Presentation
•
9th - 12th Grade

33 questions
Bonding Lesson Part 2
Presentation
•
9th - 12th Grade

31 questions
Accessing Information (Real vs Fake News)
Presentation
•
10th - 12th Grade

36 questions
2nd 6wks
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade