

Naming Acids and Bases
Presentation
•
Chemistry
•
10th Grade
•
Practice Problem
•
Medium
Standards-aligned
Jennifer Herod
Used 179+ times
FREE Resource
21 Slides • 18 Questions
1
Naming Acids and Bases

2
Acids and Bases
Acids and Bases are measured using a pH scale from 1-14. Acidic things have a low pH and Bases have a high pH. We will discuss this more in unit 9

3
Naming Acids and Bases
Acids and Bases are ionically bonded
Electrons are transferred
Naming of bases is identical to naming other ionic compounds
There are some special rules for naming acids.
4
Acids and Bases
Acids can be identified by their chemical formulas. Their chemical formulas begin with Hydrogen.
Bases can be identified by their formulas also. They end in the Polyatomic Ion, Hydroxide (OH-)
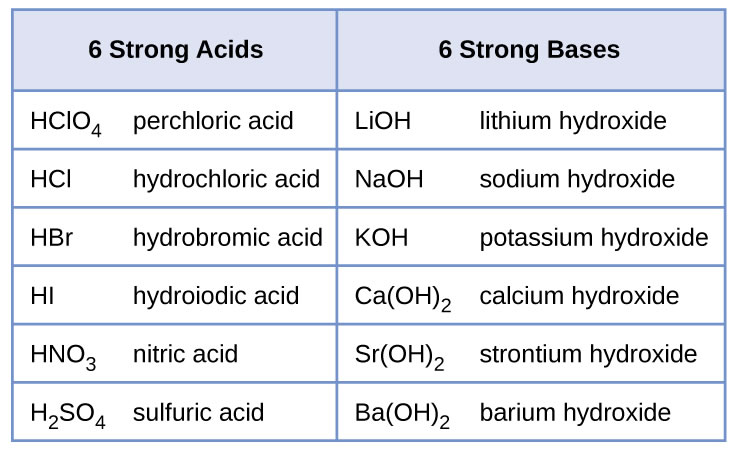
5
Acids have 2 types of naming rules
Binary
These are made of Hydrogen + element
Ternary
These are made of Hydrogen +element + Oxygen
6
Binary Acids
Hydro tells us we have a Binary Acid
The Stem indicates which element is present
Add -ic to the element stem
Add the word acid to the end.

7
Examples
HF Hydrogen and Fluorine
Hydro + fluor + ic
Hydrofluoric acid
8
Examples
HF Hydrogen and Chlorine
Hydro + chlor + ic
Hydrochloric acid
9
A few more...
HBr
Hydrobromic acid
HI
Hydroiodic acid
H2S
Hydrosulfuric acid
Note- This is different than the Ternary acid H2SO4, Sulfuric acid
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice
13
Multiple Choice
14
Ternary Acids
These are a wee bit more complicated.
First, there is no use of "Hydro" even though Hydrogen is present
Second, we must be familiar with polyatomic ions. Now is a good time to pull up your common ion chart.

15
HNO3 and HNO2
NO3- is the Nitrate ion
Looking at our chart, if the name ends in -ate, like nitrate, we change the ending to -ic
Nitrate --> Nitric + acid
HNO3 is Nitric acid

16
HNO3 and HNO2
NO2- is the Nitrite ion
Looking at our chart, if the name ends in -ite, like nitrite, we change the ending to -ous
Nitrite --> Nitrous + acid
HNO2 is Nitrous acid

17
-ate has more oxygen than -ite
H2SO4 , sulfate --> sulfuric
Sulfuric Acid -
H2SO3 sulfite --> sulfurous
Sulfurous Acid -
Phosphoric Acid - H3PO4
Phosphorous Acid - H3PO3

18
4 Variations
Hypochlorous Acid - HClO
Hypo is used to show least # of Hydrogen
Chlorous Acid - HClO2
Follows the rule
Chloric Acid - HClO3
Follows the rule
Perchloric Acid - HClO4, "Per" is added to indicate the most # of Oxygens

19
Writing formulas for Acids
Look at the element or polyatomic ion
What is the charge?
add enough Hydrogens to absorb the charge.
H+F , Fluoride ion has a 1- charge so add 1 Hydrogen, HF
H + S, Sulfur ion has a 2- charge so add 2 Hydrogens, H2S
H + P, Phosphide Ion has a 3- charge so add 3 Hydrogens, H3P
20
Polyatomic ions work the same way
PO43-, The phosphate ion, has a 3- charge so add 3 Hydrogens
H3PO4, Phosphoric acid
SO32-, the Sulfite ion, has a 2- charge so add 2 Hydrogens
H2SO3, Sulfurous acid
NO3-, the Nitrate ion has a 1- charge so add 1 Hydrogen
HNO3, Nitric acid
21
Multiple Choice
22
Multiple Choice
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
28
Multiple Choice
29
Bases
Metals ionically bonded to the polyatomic ion, Hydroxide
When writing the formula, be sure the number of Hydroxide ions matches the charge of the metal
The name is the Metal Ion + Hydroxide
Easy Peasy Lemon Squeezy

30
Base Examples
Li+ + (OH)-
Lithium charge is 1+
Hydroxide charge is 1-
We need one of each
LiOH or Li(OH)
Lithium hydroxide
31
Base Examples
Ca2+ + (OH)-
The charge of Calcium is 2+
2 Hydroxide ions are needed
Ca(OH)2
Calcium hydroxide
32
More examples
K+ + OH- -> KOH
Potassium hydroxide
Na+ + OH- -> NaOH
Sodium hydroxide, NaOH
Ba2+ + OH- --> Ba(OH)2
Barium hydroxide, Ba(OH)2
33
More examples
Rubidium hydroxide (RbOH)
Caesium hydroxide (CsOH)
Aluminum hydroxide, Al(OH)3
Strontium hydroxide, Sr(OH)2
(NH4)+ + (OH)- --> NH4OH
Ammonium Hydroxide
34
Multiple Choice
Ca(OH)2
35
Multiple Choice
36
Multiple Choice
37
Multiple Choice
38
Multiple Choice
39
Multiple Choice
Naming Acids and Bases

Show answer
Auto Play
Slide 1 / 39
SLIDE
Similar Resources on Wayground

31 questions
Chemistry CFA 2
Presentation
•
10th Grade

34 questions
Notes on Periodic Table
Presentation
•
10th - 11th Grade

30 questions
STEM Lab Safety and Procedure
Presentation
•
10th Grade

36 questions
Nitrogen Cycle
Presentation
•
9th Grade

30 questions
PAK 6 Concept 1 Review
Presentation
•
10th Grade

29 questions
Review for CA2
Presentation
•
10th Grade

28 questions
Section 2 Lesson 5-Empirical Formulas
Presentation
•
10th Grade

32 questions
A Model for Reaction Rates
Presentation
•
10th Grade
Popular Resources on Wayground

10 questions
5.P.1.3 Distance/Time Graphs
Quiz
•
5th Grade

10 questions
Fire Drill
Quiz
•
2nd - 5th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

22 questions
School Wide Vocab Group 1 Master
Quiz
•
6th - 8th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Chemistry

25 questions
Solutions Test Review
Quiz
•
10th Grade

20 questions
Acids and Bases
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

15 questions
STAAR Review Quizziz 1
Quiz
•
10th Grade

15 questions
STAAR Wayground #2
Quiz
•
10th Grade

15 questions
Ionic Compounds: Naming and Formula Writing
Quiz
•
9th - 12th Grade

10 questions
Exploring Factors Influencing Solubility
Interactive video
•
6th - 10th Grade

10 questions
Exploring the Ideal Gas Law and Its Applications
Interactive video
•
9th - 12th Grade