

Organic Chemistry
Presentation
•
Chemistry
•
10th - 11th Grade
•
Medium
nabila zulkifli
Used 86+ times
FREE Resource
10 Slides • 27 Questions
1
Organic Chemistry
C12 (Syllabus)
C10 (Coursebook)

2
Fuels
coal, natural gas & petroleum produce carbon dioxide on combustion
methane is the main constituent of natural gas
petroleum is a mixture of hydrocarbons - use fractional distillation to separate into useful fractions
3
Uses of the fractions as:
refinery gas for bottled gas for heating and cooking
gasoline fraction for fuel (petrol) in cars
naphtha fraction as a feedstock for making chemicals
diesel oil for fuel in diesel engines
bitumen for road surfaces

4

5
Multiple Choice
Petroleum is considered as a mixture because
it can be burned as a fuel.
it can be separated into fractions by distillation.
it is a fossil fuel formed over millions of years.
it is a thick, black liquid.
6
Multiple Choice

M
N
7
Multiple Choice
Fractional distillation separates crude oil based on the different __________ of the molecules in the mixture
melting point
boiling point
freezing point
chemical reactivity
8
Multiple Choice

Where do short chain hydrocarbons collect?
At the top
In the middle
At the bottom
9
Multiple Choice
Shorter hydrocarbons have:
Higher boiling points
Lower boiling points
10
Homologous series
hydrocarbon - contain hydrogen and carbon
families of compounds with the same general formula and similar chemical properties

11
Multiple Choice
Compounds in the same homologous series have
different general formulae and different chemical properties.
different general formulae and similar chemical properties.
same general formulae and different chemical properties.
same general formulae and similar chemical properties.
12
Multiple Choice
carbon only
carbon and hydrogen only
carbon, oxygen, and hydrogen.
carbon, oxygen, hydrogen, and nitrogen.
13
Multiple Choice
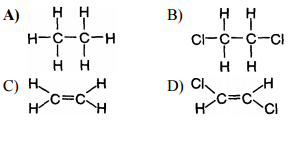
A
B
C
D
14
Alkanes
saturated hydrocarbons
contain only single covalent bonds
generally unreactive, except burning
complete combustion gives carbon dioxide and water

15
Alkenes
unsaturated hydrocarbons
contain one double covalent bond
cracking is a reaction that produces alkenes

16
Saturated vs Unsaturated
hydrocarbons:
> molecular structures
alkane - single bond only
alkene - one double bond
> reaction with aqueous bromine:
alkane - no change
alkene - yellow/orange decolourised
17
Multiple Choice
The group that has single bonds only is called as
alkenes
alcohols
alkanes
ethers
18
Multiple Choice
Which statement about alkanes is correct?
Alkanes are compounds containing carbon, hydrogen and oxygen.
Alkanes are saturated hydrocarbons.
Ethane is used to make poly(ethene).
Methane is the only alkane that does not contain a double bond.
19
Multiple Choice
Which statement about alkanes is not correct?
Alkanes are saturated hydrocarbons.
Alkanes burn to release heat energy.
Alkanes form carbon dioxide and hydrogen when they burn.
Alkane molecules contain only single bonds.
20
Fill in the Blanks
Type answer...
21
Fill in the Blanks
Type answer...
22
Multiple Choice
Orange
Blue
Colourless
Green
23
Multiple Choice
water
C4H8
C4H10
C5H8
24
Multiple Choice
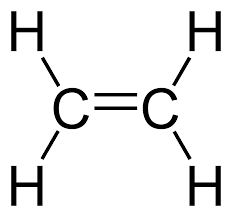
alkane
alkene
alkyne
aromatic
25
Multiple Choice

Classify the compound based on the functional group present.
alkane
alkene
alkyne
aromatic
26
Multiple Choice
20
22
40
42
27
Multiple Select
Hydrocarbon fuel + oxygen ----> ___+____
(Select 2)
CO2
O2
H2O
CxHy
28
Multiple Choice
C2H4 + 2 O2 -->
C2O2 + H4
CO2 + HOH
CO + H
2 CO2 + 2H2O
29
Multiple Choice
Which of the following reactions is/are an incomplete combustion reaction?
C5H10 + 5 O2 → 5 CO + 5 H2O
C2H4 + 3O2 → 2CO2 + 2H2O
2CH4 + 3O2 → 2CO + 4H2O
Both A and C
30
Multiple Choice
Incomplete combustion is harmful to humans because it ___________________.
increases the level of sugar in the blood
produces large amounts of carbon dioxide
decreases the amount of sugar in the blood
produces carbon monoxide that reduces the level of oxygen in the blood
31
Cracking process
> the formation of smaller alkanes, alkenes and hydrogen from larger alkane molecules
> conditions required for cracking:
high temperature (>500oC)
presence of catalyst

32
Multiple Choice
Which identifies the temperature used and describes the change to the alkane molecules during the cracking process?
100oC; the alkane molecules become larger
100oC; the alkane molecules become smaller
500oC; the alkane molecules become larger
500oC; the alkane molecules become smaller
33
Multiple Choice
Which reaction equation represents cracking?
CH4 + 2O2 → CO2 + 2H2O
C2H4 + Br2 → C2H4Br2
nC2H4 → ( C2H2 ) n
C2H6 → C2H4 + H2
34
Polymerisation
chemical process of monomers joining together to form polymers, often it takes many thousands of monomers to make a single polymer

35
Multiple Choice
What is a polymer?
a very light material that is highly flammable
a fossil fuel
a large molecule made from many monomers joined together
a molecule made up of only polar bonds
36
Multiple Choice
What is Polyethene made from?
Ethene
Ethane
Propane
Propene
37
Multiple Choice
Large molecules made up of small monomers are called
polymers
monomer
allotrope
functional group
Organic Chemistry
C12 (Syllabus)
C10 (Coursebook)

Show answer
Auto Play
Slide 1 / 37
SLIDE
Similar Resources on Wayground

31 questions
Properties of Solutions
Presentation
•
9th - 12th Grade

33 questions
Naming Covalent and Ionic Compounds
Presentation
•
9th - 12th Grade

31 questions
Classification of Matter Lesson
Presentation
•
9th - 12th Grade

34 questions
Lesson: Types of Chemical Reactions
Presentation
•
10th Grade

34 questions
6.1 Stoichiometry Pt.1 Practice
Presentation
•
10th Grade

27 questions
Subatomic Particles
Presentation
•
10th Grade

32 questions
Reaction Rates
Presentation
•
10th Grade

31 questions
Quizizz Lesson: Unit 6 Lesson 4: Covalent Naming Basics
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade