

Atomic thoeries
Presentation
•
Science
•
6th - 10th Grade
•
Practice Problem
•
Medium
+5
Standards-aligned
Divya gupta
Used 14+ times
FREE Resource
17 Slides • 35 Questions
1
Atomic thoeries

2

3
Multiple Choice
4
Multiple Choice
5
Multiple Choice
6

7
Multiple Choice

Which scientist was the first person responsible for proposing the idea of atoms?
Democritus
John Dalton
Ernest Rutherford
J.J. Thomson
Niels Bohr
8
Multiple Choice
First person to propose the name ATOMOS
Rutherford
Democritus
Dalton
Bohr
9

10
Multiple Choice
He challenged Democritus's ideas and did not believe empty space existed between atoms.
Chadwick
Bohr
Aristotle
Dalton
11
Multiple Select
According to Aristotle, the matter was made up of:
(select more than one)
Air
Earth
Fire
Water
Mud
12

13
Multiple Choice
What subatomic particle has a negative charge and is found outside of the nucleus?
Electron
Proton
Nucleus
Neutron
14
Multiple Choice

15
Multiple Choice
16
Multiple Choice
17

18
Scanning tunneling microscope:
A scanning tunneling microscope (STM) is an instrument used to see atoms by using needle on the surface of material.
19
Multiple Choice
What can a scanning tunneling microscope (STM) be used for?
Seeing objects very far away
Zooming into small objects
Creating an image of the surface of atoms
Making large objects appear small
20
Multiple Choice
Dalton did not believe that atoms could be made of smaller parts
True
False
21

22
Multiple Choice
23
Multiple Choice
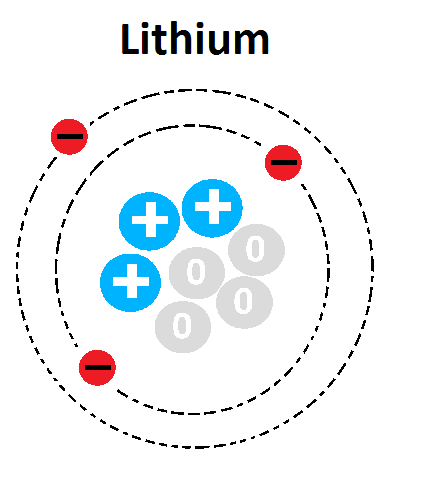
What is the mass number of this atom?
1
3
4
7
24
Atomic number:
The atomic number of an element is the number of protons found in the nucleus of every atom of that element.
25
Multiple Choice
26
Multiple Choice
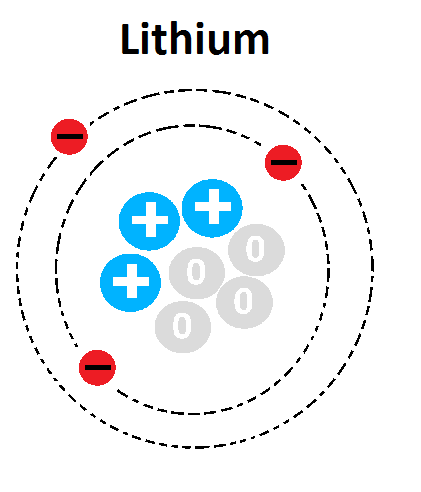
27

28

29

30
Multiple Select

What were J. J. Thomson's three contributions to the atomic theory?
Discovered the electron
Discovered the nucleus -did the gold foil experiment
Used the cathode ray tube in his discovery
Created a model of the atom with electrons moving around the nucleus in fixed orbits
Created the "plum pudding" model of the atom
31
Multiple Choice
What are electrons?
an atom’s central regions, which is made up of protons and neutrons
subatomic particles that have negative charges
subatomic particles that have neutral charges
subatomic particles that have positive charges
32
Multiple Choice
What experiment did J.J. Thomson run to develop his model of the atom?
Gold-Foil Experiment
Definite Proportion Experiment
Cathode-Ray Tube Experiment
No experimentation was done
33
Multiple Choice

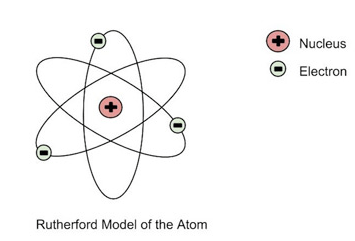
This is an example of which model of the atom?
Rutherford's Model
Plum Pudding Model
Atomic Model
Bohr's Model
34

35
Multiple Choice
What is the nucleus of the atom?
It is a centra organel of a cell
It is a center of a fruit
It is a very dense cenral core of an atom that has a positive charge
It is a very dense cenral core of an atom that has a negative charge
36

37
Multiple Choice

38
Multiple Choice

39
Multiple Choice

JJ Thomson's experiment that he used to discover electrons
gold foil experiment
cathode ray experiment
neither of these is correct
40

41

42

43
Multiple Choice
44
Multiple Choice

Rutherford's experiment that he used to discover protons and the nucleus.
gold foil experiment
cathode ray experiment
neither of these is correct
45
Multiple Choice

The Rutherford experiment concluded that
All alpha particles fired through the gold foil
The electron cloud is large and positively charged
An atom barely has any empty spaces
The nucleus is tiny and heavy compared to the rest of an atom
46
Multiple Choice

47
Multiple Choice
Why did Ernest Rutherford and his colleagues perform the Gold-Foil Experiment?
They wanted to test and confirm the Plum-Pudding model
They wanted to test John Dalton's Model
They were looking for electrons
They wanted to come up with their own Atomic Model
48
Multiple Choice
Based on Rutherford's model of the atom, electrons in the atom are located
inside of the nucleus of the atom
outside and around the nucleus of the atom
outside and inside of the nucleus of the atom
49
Multiple Choice
What results did Rutherford observe in his Gold-Foil experiment?
He observed that all alpha particles did go through a gold-foil in straight lines
He observed that most alpha particles did not go through a gold-foil and stopped on one side of the gold foil.
He observed that a small number of alpha particles bounced off the gold-foil at very large angles
He observed that all alpha particles slightly deflected from the straight line when going through a gold-foil
50
Multiple Choice
51
Multiple Choice
Which of the following is/are conclusions based on Rutherford’s gold foil experiment?
Atom is mostly empty space
The nucleus is positively charged
The atom has a small dense nucleus
All answers are correct
52
Multiple Choice
Which of the following is the best evidence that the particles that make up a cathode ray had a negative charge?
The particles were produced by many different metals.
The ray was deflected toward a positive electric field.
Line emission spectra for an element are always the same.
The ray caused a phosphorescent screen to glow.
Atomic thoeries

Show answer
Auto Play
Slide 1 / 52
SLIDE
Similar Resources on Wayground

44 questions
Symbiosis Lesson
Presentation
•
6th - 10th Grade

47 questions
Elkhorn History Images
Presentation
•
KG

43 questions
Fossil Fuels
Presentation
•
5th - 9th Grade

47 questions
Understanding Plate Tectonics
Presentation
•
6th - 8th Grade

46 questions
Review for Unit Test #3
Presentation
•
6th - 9th Grade

52 questions
Our Solar System - Overview
Presentation
•
7th - 10th Grade

45 questions
Physical Science Motion Lesson
Presentation
•
6th - 9th Grade

46 questions
Homeostasis & Feedback
Presentation
•
9th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

20 questions
Rocks and The Rock Cycle
Quiz
•
6th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

15 questions
Punnett Squares
Quiz
•
6th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

18 questions
Oceans, Atmosphere, Climate Vocabulary Amplify
Quiz
•
6th Grade