

Final Exam Review
Presentation
•
Chemistry
•
11th - 12th Grade
•
Medium
+2
Standards-aligned
Hector Mendoza-Arias
Used 4+ times
FREE Resource
19 Slides • 12 Questions
1
Final Exam Review 1.1

2
CHM 1.1
Chm.1.1.1 Analyze the structure of atoms, isotopes, and ions.
Chm.1.1.2 Analyze an atom in terms of the location of electrons.
Chm.1.1.3 Explain the emission of electromagnetic radiation in spectral form in terms of the Bohr model.
Chm.1.1.4 Explain the process of radioactive decay by the use of nuclear equations and half-life.
3
Chm.1.1.1 Analyze the structure of atoms, isotopes, and ions.
Atoms
Isotopes
Ions

4
Atoms
Protons: 1 amu
Neutrons: 1 amu
Electrons: 1/2000 amu

5
Atomic # and Mass
Atomic Number Represented by "Z"
Atomic Mass Represented by "A"

6
Multiple Choice
the central region of an atom where its neutrons and protons are is its
nucleus
electron cloud
core
center
7
Isotopes
different forms of the same element that contain equal numbers of protons but different numbers of neutrons

8
X(element symbol) -- A (Mass Number)
Another way of writing isotopes
9
Average Atomic Mass vs Isotopic Mass vs Mass Number
AAM-the sum of the masses of its isotopes, each multiplied by its natural abundance
IM- mass of the most abundant isotope of an element
MN- the total number of protons and neutrons in a nucleus.

10
Multiple Choice
11
Multiple Choice
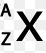
12
Multiple Choice

How many neutrons does the isotope of lithium have?
8
3
4
5
13
Multiple Choice
14
Multiple Choice

What does the 1.00794 stand for?
mass number
atomic number
average atomic mass
number of protons
15
Average Atomic Mass
The average atomic mass of an element can be found on the periodic table, typically under the elemental symbol.
Average atomic mass = f1M1 + f2M2
f=fraction representing the natural abundance of the isotope
M= the mass number (weight) of the isotope.

16
Multiple Choice
17
Ions
an atom or molecule with a net electric charge due to the loss or gain of one or more electrons.

18
Multiple Choice
19
Chm.1.1.2 Analyze an atom in terms of the location of electrons.
Bohr Model of the Hydrogen Atom
Electron Clouds

20
Bohr's Model of the Hydrogen Atoms
electrons can be moved into different orbits with the addition of energy
When the energy is removed, the electrons return back to their ground state, emitting a corresponding amount of energy – a quantum of light, or photon(quantum theory)

21
Electron Cloud Model
We can not know exactly where an electron is at any given time, but the electrons are more likely to be in specific areas.

22
Multiple Choice
23
Chm.1.1.3 Explain the emission of electromagnetic radiation in spectral form in terms of the Bohr model.
Quanta
Excited and Ground States
Emissions Spectrum

24
Bohrs Model of an Atom
An electron circles the nucleus only in fixed energy ranges called orbits.
An electron can neither gain nor lose energy inside this orbit, but could move between orbits by absorbing or releasing a specific amount of energy.
The lowest energy orbit is closest to the nucleus.

25
Quantum
Energy exists in discrete units called quanta.(the minimum amount of energy that can be absorbed or released by matter)

26
Ground State Vs Excited State
Ground State: the lowest possible energy that an atom can have
Excited State: energy level of an atom, ion, or molecule in which an electron is at a higher energy level than its ground state.

27
Multiple Choice
28
Multiple Choice
Ground state means that an electron is...
at its lowest possible potential energy
in the first shell of an atom
laying low for the weekend
removed from an atom
29
Multiple Choice
Ground state means that an electron is...
at its lowest possible potential energy
in the first shell of an atom
laying low for the weekend
removed from an atom
30
Emissions Spectrum
When the electron moves to a lower energy level, it releases the energy difference in the two levels as electromagnetic radiation

31
Electromagnetic Radiation
this electromagnetic radiation is given off as photons(a particle representing a quantum of light).
Look at your Chem Reference Tables

Final Exam Review 1.1

Show answer
Auto Play
Slide 1 / 31
SLIDE
Similar Resources on Wayground

25 questions
BODY LANGUAGE
Presentation
•
11th - 12th Grade

26 questions
The Metric System Basics
Presentation
•
11th - 12th Grade

25 questions
Neutralizations and Titrations
Presentation
•
10th - 12th Grade

23 questions
Periodic Trend Practice
Presentation
•
10th - 12th Grade

24 questions
Matter-States and Changes
Presentation
•
11th - 12th Grade

25 questions
Probability of Simple Events
Presentation
•
11th - 12th Grade

23 questions
Direct Object Pronouns
Presentation
•
11th - 12th Grade

25 questions
Covalent Bonds
Presentation
•
10th - 11th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Redox Reactions
Quiz
•
10th - 12th Grade

20 questions
pH and pOH Calculations
Quiz
•
10th - 11th Grade

36 questions
Unit 4 (Part 2) Solutions
Quiz
•
10th - 12th Grade

30 questions
Final Exam Prep S22: 2.1.A.1 Isotopes
Quiz
•
9th - 12th Grade

25 questions
Stoichiometry Mass to Mass
Quiz
•
10th - 11th Grade

15 questions
Calculating pH and pOH
Quiz
•
10th - 12th Grade

15 questions
Balancing Chemical Equations
Quiz
•
10th - 12th Grade