

LESSON - 3rd 6wks
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
+3
Standards-aligned
Kyle Pearson
Used 14+ times
FREE Resource
18 Slides • 30 Questions
1
3rd 6wks Review
Electromagnetic Spectrum
Periodic Trends
Atomic Theory
Classifying Reactions
Balancing Chemical Equations
Molar Mass
Moles
Percent Composition Cm
2

3

4
Multiple Choice
Select the order of waves from long wavelength to short wavelength on the EMS
5



6
Multiple Choice
7
Multiple Choice
8
Multiple Choice

As you move down a GROUP on the periodic table the atomic radius get bigger. This is because ____________.
The atoms have more neutrons in the nucleus.
The atoms have more protons in the nucleus.
The atoms have more energy levels for the electrons
The atoms have greater repulsion forces between the protons and electrons
9
Multiple Choice
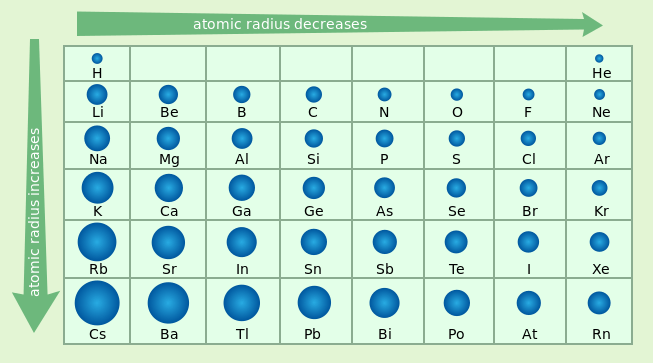
As you move across a period on the periodic table the atomic radius tend to get smaller because, ______________.
the atoms have more mass making them heavier.
more neutrons, making the nucleus bigger.
the atoms have more protons, pulling the electrons closer to the nucleus
the atoms have less energy levels for the electrons to occupy.
10
Multiple Select
Where is electronegativity and ionization energy the highest on the Periodic Table?
Click all that apply
up the group
down the group
left to right
right to left
11

12
Scientists of the Atomic Theory
Democritus
everything in the world is made of tiny particles called atomos,
surrounded by empty space.
Aristotle
believed that matter was made of 4 elements, earth, wind, water, and fire.
13
Scientists of the Atomic Theory
J.J. Thomson
won a Noble Prize for his discovery of electrons
Ernest Rutherford
is considered the Father of the Nuclear Age with his Gold Foil
experiment that discovered the nucleus
14
Scientists of the Atomic Theory
John Dalton's Postulates
ALL matter is made of tiny atoms
Atoms that are the same are identical in every aspect
Atoms CANNOT be divided, they are indivisible
Atoms CANNOT be CREATED or DESTROYED
Atoms combine to form compounds
In chemical reactions atoms are combined, separated, and rearranged
15
Scientists of the Atomic Theory
Werner Heisenbery
came up with the Uncertainty Principle that says it is impossible to determine the exact position and speed of an electron when it is orbiting the nucleus.
Niels Bohr's
theory was that electrons moved around the nucleus at fixed energies
and distances.
16
Multiple Select
John Dalton stated:
click ALL that apply
Atoms are tiny, invisible particles.
Atoms of one element are all the same.
Atoms of different elements are different.
Compounds form by combining atoms.
Atomos means indivisible
17
Multiple Choice
He claimed that matter was made of small, hard particles that he called “atomos.”
Democritis
Dalton
Bohr
Einstein
18
Multiple Choice

Which Scientist created this model and
proposed the theory that atoms orbit the nucleus in specific orbits.
Democritus
Dalton
Bohr
Rutherford
19
Multiple Select
Rutherford is know for ......
Gold foil experiment
Noble Prize for finding electrons
discovering the nucleus
Father of the Nuclear Age
20
Multiple Choice
JJ Thomson won the Noble Peace prize for discovering what subatomic particle?
nucleus
neutron
proton
electron
21
Multiple Select
Aristotle believed that matter was made of what 4 elements?
22

23

Why are reactions balanced?
24
Fill in the Blanks
Type answer...
25
Multiple Choice
Matter has to be balanced or the chemical equation will blow up
26
Multiple Select
Identify the REACTANTS :
H2O + CO2 → HCO + HO
carbon dioxide
hydrogen monoxide
dihydrogen monoxide
carbonic aicd
27
Fill in the Blanks
Type answer...
28
Fill in the Blanks
Type answer...
29
Fill in the Blanks
Type answer...
30
Multiple Choice
31
Fill in the Blanks
Type answer...
32
Fill in the Blanks
Type answer...
33
Fill in the Blanks
Type answer...
34

35
Multiple Select
Click all the things you can use to balance a chemical equation
coefficient
subscripts
superscripts
chemical symbols
36
Multiple Select
Find the ERROR(s) in the decomposition reaction and replace them with the correct information.
4H2(g) + 2O2(g) >> 4H2O(l)
it is not balance
coefficients need to be reduced
the (g) and (l) are in the wrong place
decomposition reaction
combination reaction
37
Multiple Select
Identify the letter in the parenthesis
4H2(g) + 2O2(g) >> 4H2O(l)
(g) gas
(g) gel like
(l) liquid
(l) light gas
(l) Iodine
38

39
Mole Concept
Mole concept is used to help chemists count very small particles
Avogadro's Number = 6.02e23
Molar mass (g/mol) is the mass of one mole of substance
example - molar mass of water is 18 g/mol
40
Conversion Factors
1 mole particles = 6.02e23 particles
1 mole particles = molar mass (g) from Periodic Table
1 molar particles = 22.4 liters (volume)
1 molecule = # atoms in that molecule
41
Steps to follow to solve Mole Problems
Identify the GIVEN and what the problem is asking you to convert to
Diagonal Rule - SAME UNIT (words ONLY)
Find the other half your conversion factor
Round the answer using sig figs from the GIVEN
42
Fill in the Blanks
Type answer...
43
Multiple Choice
Ammonium nitrate, NH4NO3, commonly known as ANFO, is a compound used to make explosive devices. How many grams are in one mole of ammonium nitrate to the nearest thousandths place?
31.014 g
45.021 g
48.045 g
80.043 g
44
Multiple Choice
If one Monster energy drink contains 0.16 g of caffeine, how many moles of caffeine do we consume when we drink 1 can of Monster?
The chemical formula for caffeine is C8H10N4O2 and the molar mass is 194.
194.19 moles
1.07 moles
0.00082 moles
1213.69 moles
45
Multiple Choice
A student needs 65.0 g of copper(II) chloride for an experiment.
How many atoms of copper is this?
2.91e23 atoms
3.91e25 atoms
2.35e23 atoms
4.70e23 atoms
46
% Composition
Hopefully you watched the lesson and have practiced!
47

48
Fill in the Blanks
Type answer...
3rd 6wks Review
Electromagnetic Spectrum
Periodic Trends
Atomic Theory
Classifying Reactions
Balancing Chemical Equations
Molar Mass
Moles
Percent Composition Cm
Show answer
Auto Play
Slide 1 / 48
SLIDE
Similar Resources on Wayground

41 questions
Arrhenius & Bronsted Lowry Acids & Bases
Presentation
•
9th - 12th Grade

43 questions
4/22 Solutions in Chemistry
Presentation
•
9th - 12th Grade

45 questions
(Full Lesson) Biology EOC Reporting Category 5 Review
Presentation
•
9th - 12th Grade

44 questions
Introduction to Moles
Presentation
•
9th - 12th Grade

41 questions
Covalent Bonding
Presentation
•
8th - 12th Grade

41 questions
Lewis Dot Structures
Presentation
•
9th - 12th Grade

42 questions
Chapter 13 Vocab Review - Thermochemistry
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

25 questions
Waygrounds 5/20 PS.8.1.2
Quiz
•
9th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

15 questions
Types of chemical reactions
Quiz
•
9th - 12th Grade

32 questions
Acids/Bases, Electrochem, Orgo
Quiz
•
9th - 12th Grade