

Advanced Mole Conversions
Presentation
•
Chemistry
•
10th Grade
•
Practice Problem
•
Medium
Standards-aligned
Joshua Baltzell
Used 150+ times
FREE Resource
7 Slides • 6 Questions
1
Advanced Mole Conversions
This lesson is designed as an extension to show you more advanced mole conversion formats. This will help you as we get deeper into stoichiometry.
2
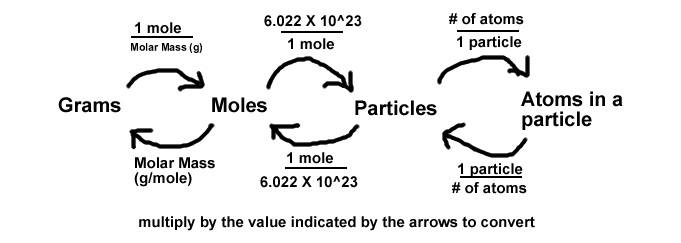
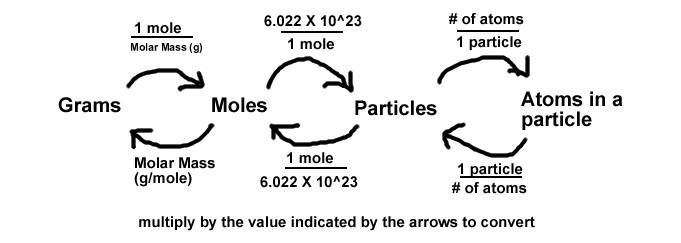
Review the Basics
First, let's review some of the basics so we know what we're covering.
Whenever working with mass, moles, or particles, we can always convert between them for any element or compound by using the conversion factors molar mass and Avogadro's number.
g/mol
3
Example
If I have 4.3 moles of Lithium, how many grams of Lithium is that?
4
Multiple Choice
You try, just to make sure you're ready to move on to this level of material:
If you had 3 moles of Sodium, how many grams of sodium would you have?
68.97 g Na
7.663 g Na
0.1305 g Na
1.807E24 atoms Na
5
Moles within Compounds
When looking at individual compounds, we can see the number of atoms used to construct any compound. Since atoms are directly transferable to moles, we can convert between moles of an atom to moles of a compound using mole to mole ratios.
It takes 2 moles of hydrogen and 1 mole of oxygen to make 1 mole of dihydrogen monoxide. Each of these numbers creates a conversion factor.
6
Mole to Mole Conversions
The image shows us multiple ratios we can make based on the formula for water. We can see that for every 2 moles of hydrogen we have, there will be 1 mole of oxygen.
Therefore, if I had 7 moles of hydrogen, I could calculate the number of moles of oxygen: 3.5 (Remember that we can have decimals for moles, but not for atoms).

7
Multiple Choice
For the compound Fe2O3, if we had 4 moles of Iron (Fe), how many moles of oxygen would we have?
6 moles O
3 moles O
4 moles O
9 moles O
8
Multiple Choice
For the compound MgF2, if we had 6 moles of fluoride, how many moles of the entire compound would we have?
3 moles MgF2
6 moles MgF2
1 mole MgF2
2 moles MgF2
9
This is an intermediate step...
We can use this to convert between elements and compounds now!
Say I had 50 grams of H2O, and wanted to know how many grams of Oxygen this was.
I could spend time using the Law of Definite proportions to find the proportional percent of each element, or I could just convert (which is different steps but it mathematically doing the same thing).
10
Example
Using the conversion factors that I already know, and the ones we just learned, I can set up my railroad tracks to calculate the mass of pure oxygen in 50 grams of H2O.
This will be helpful for when I want to use an element to create a compound, or when I want to find details about an element within a compound.

11
Multiple Choice

For the compound NaCl, if we had 10 grams of Chlorine (Cl), how many moles of Sodium would we have?
0.282 mol Na
3.55 mol Na
35.45 mol Na
1.55 mol Na
12
Multiple Choice
If I had 17.0 grams of Hydrogen (H) in the compound HCl, how many grams of Chloride (Cl) would we have?
597.9 g Cl
0.4757 g Cl
46.23 g Cl
2.563 g Cl
13
Multiple Choice

For the compound AgNO3, it took
5.873⋅1024 atoms of oxygen (O) to make the compound. How many grams of silver (Ag) was needed to make the compound?Hint: If you need help setting up the problem, check the image.
350.8 g Ag
1052.3 g Ag
107.9 g Ag
2.11⋅1026 g Ag
Advanced Mole Conversions
This lesson is designed as an extension to show you more advanced mole conversion formats. This will help you as we get deeper into stoichiometry.

Show answer
Auto Play
Slide 1 / 13
SLIDE
Similar Resources on Wayground

9 questions
Arts and Crafts
Presentation
•
KG - 10th Grade

11 questions
PSAT Orientation
Presentation
•
10th Grade

10 questions
ENLACE QUÍMICO
Presentation
•
1st - 10th Grade

9 questions
II° Síntesis de Unidad I
Presentation
•
10th Grade

7 questions
Lessons 11-12 Career planning
Presentation
•
10th Grade

10 questions
UAS P3KIMIA
Presentation
•
10th Grade

10 questions
Intermolecular Forces
Presentation
•
10th Grade

10 questions
Percent comp, empirical, molecular formula
Presentation
•
10th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

15 questions
Nuclear Chemistry: Balancing
Quiz
•
10th Grade

20 questions
Naming Acids and Bases
Quiz
•
10th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade