

PS - Matter: Properties and Changes
Presentation
•
Science, Chemistry
•
8th - 11th Grade
•
Practice Problem
•
Medium
Nichole SouthGarnerHS
Used 28+ times
FREE Resource
13 Slides • 70 Questions
1
Physical Science Finals Review #1 - Matter: Properties and Changes
In this review, we will look at Types of Matter, Phases of Matter, Properties of Matter, and the Structure of Matter.

2

3

4
Multiple Choice

How do you identify a compound?
Same type of atoms and molecules
Same type of atoms different type of molecules
Different type of atoms same type of molecules
Different type of atoms and molecules
5
Multiple Choice

What type of matter is this?
Element
Compound
Mixture of elements
Mixture of compounds
6
Multiple Choice

What type of matter is this?
Compound
Element
Mixture of elements
Mixture of compounds
7
Multiple Select
Which of the following are compounds?
Silver nitrate (AgNO3)
Bronze (Cu + Sn)
Sugar (C12H22O11)
Blood
8
Multiple Select
Which of the following are mixtures?
Sodium bicarbonate (NaHCO3)
Steel (Fe + C)
Salt water (NaCl + H2O)
Gold (Au)
9
Multiple Choice
What type of mixture is air (with smog) ?
Homogeneous
Heterogeneous
10
Multiple Select
Which of the following are pure substances?
Carbon dioxide (CO2)
Milk
Salt water
Nitrogen (N2)
11

12

13
Multiple Choice
Solubility refers to the ____ of solute that can dissolve in a certain volume or mass of solvent, at a certain temperature.
Mean
Range
Amount
Mode
14
Multiple Choice
15
Multiple Choice

This type of mixture contains two or more substances that are visibly distinguishable.
homogeneous
heterogeneous
solution
colloid
16
Multiple Choice
Kool-Aid - Powder, sugar, and water
Identify the solvent
water
powder
sugar
powder and sugar
17
Multiple Choice
This is the part of a solution that dissolves
Solute
Solvent
Solution
Mixture
18
Multiple Choice
What is a solvent?
The substance that does the dissolving in a solution.
The substance that is being dissolved in a solution.
The mixing of different substances.
The process in which neutral molecules lose or gain electrons
19
Multiple Choice
20
Multiple Choice
21
Multiple Choice
22

23
Multiple Choice
24
Multiple Choice
The type of matter with the lowest heat energy and temperature.
gas
solid
liquid
plasma
25
Multiple Choice
26
Multiple Choice
Liquids and gases are similar because __________________________.
they both take the shape of their container
they both have their own shape
liquids pour and gases don't
liquids take the shape of the container but gases have their own shape
27
Multiple Choice
28
Multiple Choice
What Is Matter?
Anything That has mass and takes up space.
Anything that you can see or taste.
What you can touch.
you and go throught this.
29
Multiple Choice
Which state of matter has a definite shape?
solid
liquid
gas
plasma
30
Multiple Choice
The state of matter where particles are very spread out and moving quickly.
gas
liquid
solid
Helium
31
Multiple Choice
The state of matter that has particles close together and vibrating.
liquid
solid
gas
water
32

33
Multiple Choice
Water vapor changing directly into a solid is called
condensation
evaporation
sublimation
deposition
34
Multiple Choice
35
Multiple Choice
36
Multiple Choice

The drops of water that appear on the outside of a glass of cold juice on a warm day are an example of?
sublimation
condensation
evaporation
37
Multiple Choice
38
Multiple Choice
39
Multiple Choice
40
Multiple Choice

41

42
Multiple Choice
Which process is an example of of a physical change?
ice melting
milk souring
metal rusting
wood burning
43
Multiple Choice
Which of these choices describes a chemical property?
flexibility
boiling point
reactivity with water
electrical conductivity
44
Multiple Choice
Which of these choices is an example of a physical property?
the tarnishing of silverware
the texture of a piece of chocolate
the effect of acid rain on automobiles
the combustion of gasoline in a car engine
45
Multiple Choice
Which of these best describes physical properties?
Physical properties behave identically for all matter under the same conditions.
Physical properties can be observed without changing the identity of a substance.
Physical properties include flammability.
Physical properties cause atoms and molecules to change structure when substances are mixed.
46
Multiple Choice
Which process is a sign of a chemical change?
a rotting potato gives off a bad smell
a melting block of ice leaves a large puddle
a cloud quickly changes shape when blown by the wind
a plaster statue breaks when it falls on the floor
47
Multiple Choice
Which of these choices is a physical property?
solubility
flammability
ability to rust
reaction with water
48

49
Multiple Choice
What about gases can be measured?
Pressure and Volume
Temperature, Volume, and Pressure
Volume and Temperature
Pressure, Temperature, Volume, and Moles
50
Multiple Choice
If a hairspray can is heated, what can be expected of the pressure of the gas inside the can?
The pressure will increase
The pressure will decrease
The pressure will remain constant
The pressure will equalize
51
Multiple Choice
A student inflates a balloon with helium then places it in the freezer. The student should expect
the balloon's volume to increase
the balloon's volume to decrease
the balloon's moles to increase
the balloon's moles to decrease
52
Multiple Choice
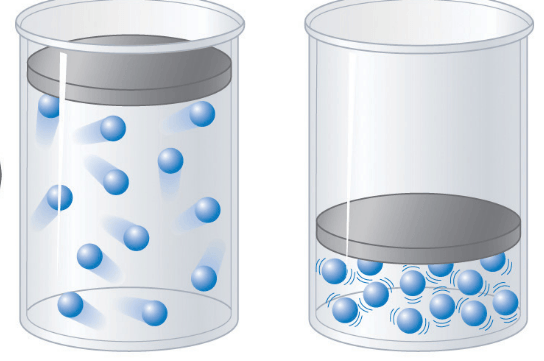
53
Multiple Choice
54
Multiple Choice
55
Multiple Choice
56

57
Multiple Choice

What is the atomic mass of "F"
9
18
17
19
58
Multiple Choice

What is the number of protons that the element in this image contain?
14
7
15
18
59
Multiple Choice

How many neutron are in the atom "K"?
7
2
39
12
60
Multiple Choice
Which particle has a similar mass to a proton?
Electron
Neutron
Photon
Marble
61
Multiple Choice
62
Multiple Choice
63
Multiple Choice
64
Multiple Choice
65
Multiple Choice
66
Multiple Choice
67
Multiple Choice
68
Multiple Choice
69
Multiple Choice
Particles in an atom that are neutral and have no charge are
negatrons
electrons
neutrons
protons
70
Multiple Choice
Particles in an atom that are neutral and have no charge are
negatrons
electrons
neutrons
protons
71

72

73

74
Multiple Choice
75
Multiple Choice

How many valence electrons does Aluminum Have?
1 Valence electron
2 Valence electron
3 Valence electron
4 Valence electron
5 Valence electron
76
Multiple Choice

How many valence electrons does Nitrogen Have?
1 Valence electron
2 Valence electron
3 Valence electron
4 Valence electron
5 Valence electron
77
Multiple Choice

What is the correct lewis dot diagram for Boron?
78
Multiple Choice
What is a Bohr Model?
a simplified representation of an atom
vertical column in periodic table
horizontal row in periodic table
Only shows the element symbol and it's outer most electron shell
79
Multiple Choice
How many electrons can go in the first energy level?
2
8
18
32
80
Multiple Choice

81
Multiple Choice
Which Bohr model represents Neon?
82
Multiple Choice

83
Multiple Choice
Physical Science Finals Review #1 - Matter: Properties and Changes
In this review, we will look at Types of Matter, Phases of Matter, Properties of Matter, and the Structure of Matter.

Show answer
Auto Play
Slide 1 / 83
SLIDE
Similar Resources on Wayground

75 questions
Skeletal SYS
Presentation
•
9th - 12th Grade

82 questions
Scientific notation
Presentation
•
7th - 12th Grade

75 questions
Bone Formation
Presentation
•
9th - 12th Grade

77 questions
Introduction to Biochemistry Lesson
Presentation
•
9th - 12th Grade

78 questions
2.1 Matter Notes
Presentation
•
9th - 12th Grade

80 questions
APES Unit 2 Videos Biodiversity
Presentation
•
9th - 12th Grade

80 questions
Flow of Energy and Matter
Presentation
•
9th - 12th Grade

76 questions
Pre AP Biology Natural Selection and Hardy Weinberg
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

40 questions
NCFE Earth and Environmental Science Released Test
Quiz
•
9th - 12th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

19 questions
Renewable and Nonrenewable Resources (CK)
Quiz
•
8th Grade

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade