

Introduction to Biochemistry Lesson
Presentation
•
Science, Biology
•
9th - 12th Grade
•
Practice Problem
•
Medium
+10
Standards-aligned
David Blankenship
Used 21+ times
FREE Resource
23 Slides • 54 Questions
1
Introduction to Biochemistry Lesson

2

3

4
Open Ended
What is your definition of the word biochemistry?
5
Multiple Choice
How do organic compounds differ from inorganic compounds?
All organic compounds contain carbon.
Only organic compounds contain hydrogen.
Organic compounds are natural, while inorganic compounds are synthetic.
Organic compounds contain water, while inorganic compounds do not contain water.
6

7

8

9

10

11

12
Multiple Choice

Which picture represents an element/molecule made of the same atoms?
O2
H2O
13
Multiple Choice

14
Multiple Choice

What do elements in the same period have in common with each other?
Same number of electrons.
Same number of energy levels (shells).
Same properties.
Same number of protons.
15
Multiple Choice
What is matter?
Volume and color
Volume and density
Volume and length
Volume and mass
16
Multiple Choice
17
Multiple Choice
18
Multiple Choice

What do elements in the same group (family) have in common with each other?
Properties and valence electrons
Atomic number and valence electrons
Chemical symbol and atomic mass
Atomic mass and atomic number
19
Multiple Choice
How are the elements arranged on the periodic table?
Atomic number
Atomic mass
Alphabetically
Chemical symbol
20
Multiple Choice

21
Multiple Choice

22
Multiple Choice
23
Multiple Choice
The smallest part of any substance is?
Element
Compound
Atom
Molecule
24
Multiple Choice
A substance that cannot be separated or broken down into simpler substances by chemical means is?
Mixture
Element
Compound
Molecule
25

26
Multiple Choice

Select the correct answer.
The first model represents the proton and neutron distribution in the nucleus of four helium isotopes. The second image shows the entry for helium in the periodic table.
Based on the information in the periodic table, which is the most common isotope of helium?
27

28

29
Multiple Choice
Where are electrons found?
the nucleus
next to the neutrons
the proton
orbiting the nucleus
30
Multiple Choice
What is an ion?
a charged atom
an atom with different neutrons
atoms with no protons
atoms with no electrons
31
Multiple Choice
What is the chemical symbol for the element Carbon?
C
Ca
Carb
CO
32
Multiple Choice
Which of the following is a chemical symbol?
H2O
O
NaCl
FeO
33
Multiple Choice
Which of the following is a chemical formula?
H2SO4
Si
Hg
C
34
Multiple Choice
What is the neutron?
a negatively charged particle that orbits the nucleus
a neutral particle found in the nucleus
a positively charged particle found in the nucleus
a neutral particle that orbits the nucleus
35
Multiple Choice
What is the electron?
a negatively charged particle that orbits the nucleus
a neutral particle found in the nucleus
a positively charged particle found in the nucleus
a neutral particle that orbits the nucleus
36
Multiple Choice
What is the proton?
a negatively charged particle that orbits the nucleus
a neutral particle found in the nucleus
a positively charged particle found in the nucleus
a neutral particle that orbits the nucleus
37
Multiple Choice
What two subatomic particles are found in the nucleus?
proton and electron
electron and neutron
neutron and proton
neutron and electron
38
Multiple Choice
What is a compound?
Two different elements bonded together.
Two of the same elements bonded together.
A mixture of different substances.
Two substances stuck together with glue.
39
Multiple Choice
What is a molecule?
two elements bonded together (sharing electrons)
a mixture of different substances
smallest individual particles
salt mixed in water
40

41

42
Multiple Choice
This type of bonding can be described as "I give, I get"
ionic
covalent
metallic
43
Multiple Choice
This type of bonding can be described as a "cooperation"
ionic
covalent
metallic
44
Multiple Choice
What type of bond involves the sharing of electrons between atoms?
covalent bond
ionic bond
metallic bond
transfer bond
45
Multiple Choice
A bond between a nonmetal and a nonmetal is called a(n)
covalent bond
ionic bond
metallic bond
transfer bond
46
Multiple Choice
A bond between a metal and a nonmetal is called a(n)
covalent bond
ionic bond
metallic bond
transfer bond
47

48

49
Multiple Choice
50
Multiple Choice
51
Multiple Choice
52
Multiple Choice
Al + O2 --> 2Al2O3
53
Multiple Choice
54
Multiple Choice
55
Multiple Choice
2H2 + O2 --> 2H2O
56
Multiple Choice
2H2 + O2 --> 2H2O
57
Multiple Choice
58

59

60

61

62

63

64
Multiple Choice
Polymer's split apart to form ________, by __________.
monomers, hydrolysis
monomers, dehydration synthesis
amino acids, hydrolysis
amino acids, dehydration synthesis
65
Multiple Choice
66
Multiple Choice
67
Multiple Choice

68
Multiple Choice

69
Multiple Choice

70
Multiple Choice

71
Multiple Choice
72
Multiple Choice

73
Multiple Choice

74
Multiple Choice
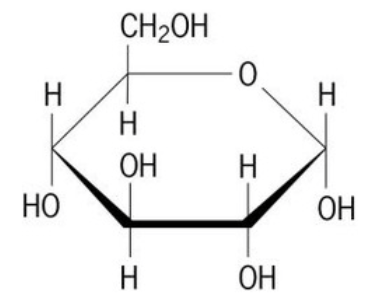
75
Multiple Choice
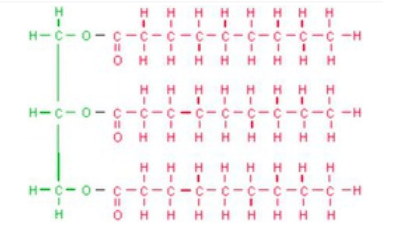
76

77
Open Ended
Some of the most promising treatments for cancer are targeted therapies.
Use information from credible websites to explain how targeted cancer therapies differ from traditional chemotherapies, and describe why these therapies are an advancement in medicinal chemistry.
Introduction to Biochemistry Lesson

Show answer
Auto Play
Slide 1 / 77
SLIDE
Similar Resources on Wayground

70 questions
Phylum Mollusca Notes
Presentation
•
10th - 12th Grade

72 questions
Senderos 2 Lección 4 - El Cuerpo
Presentation
•
10th - 12th Grade

74 questions
Chemistry 1st Quarter Review: Eve
Presentation
•
9th - 12th Grade

70 questions
TSIA2 Boot Camp - Unlocking Word Meaning
Presentation
•
9th - 12th Grade

75 questions
Skeletal SYS
Presentation
•
9th - 12th Grade

69 questions
5.1.5 Life Science Practice
Presentation
•
9th - 12th Grade

69 questions
Physical and Chemical Properties
Presentation
•
9th - 12th Grade

75 questions
Bone Formation
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade