

Mole Conversions
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
Standards-aligned
Daven Vandenberg
Used 230+ times
FREE Resource
5 Slides • 13 Questions
1
Mole Conversions

2

3
Multiple Choice
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice
Which of the following statements regarding the mole is INCORRECT?
A mole is a unit of quantity equal to 6.02 x 1023 particles.
The number of particles in a mole is known as Avogadro’s number.
A mole of particles of an element is numerically equal to the atomic mass of the element.
none of the above
8

9
Multiple Choice
10
Multiple Choice
11
Multiple Choice
How many molecules are in 2.5 mol of NaCl?
1.51x1023
146
4.15
1.51x1024
12

13
Multiple Choice
In order to convert from grams of a substance to moles of a substance, I would need to ___________.
divide by the molar mass
divide by the molecular mass
divide by the molecular weight
divide by the mass of one mole
14
Multiple Choice
What is the molar mass of Calcium Oxide (CaO)?
56.1
112.2
24.0
40.1
15
Multiple Choice
How many moles are equal to 89.23 g of calcium oxide, CaO?
1.59 mol
2.03 mol
1.77 mol
3.21 mol
16
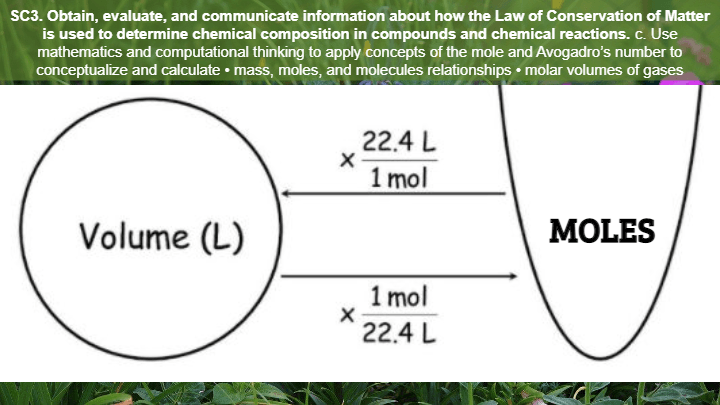
17
Multiple Choice
At STP, 1 mol of gas has a volume of
34L
22.4L
60L
0.6L
18
Multiple Choice
Determine the volume, in liters, of 1.2 mole SO2 gas at STP.
13 L
26 L
6.5 L
Mole Conversions

Show answer
Auto Play
Slide 1 / 18
SLIDE
Similar Resources on Wayground

13 questions
Extensive & Intensive Physical Properties
Presentation
•
10th - 12th Grade

13 questions
Properties of Parallelograms
Presentation
•
9th - 12th Grade

13 questions
Types of Chemical Reactions
Presentation
•
9th - 12th Grade

14 questions
Percent Yield
Presentation
•
9th - 12th Grade

14 questions
Balancing Chemical Equations Review
Presentation
•
9th - 12th Grade

13 questions
SI Unit Prefix Conversions
Presentation
•
9th - 12th Grade

11 questions
Balancing, Conservation and Reaction Types Review
Presentation
•
9th - 12th Grade

16 questions
yr 9 chemistry revision-separation techniques
Presentation
•
9th - 11th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade