

Naming, Percent Composition, Matter, Atoms
Presentation
•
Chemistry
•
10th Grade
•
Practice Problem
•
Easy
+1
Standards-aligned
Tyler Jones
Used 9+ times
FREE Resource
1 Slide • 29 Questions
1
Naming, Percent Composition, Matter, Atoms

2
Multiple Choice
If carbon-12 had one less proton, it would become...
boron-11
nitrogen-11
carbon-11
carbon-13
3
Multiple Choice
If carbon-12 had one less neutron, it would become...
boron-11
nitrogen-11
carbon-11
carbon-13
4
Multiple Choice
If carbon-12 had one less electron, it would become...
isotope
cation
anion
neutral atom
5
Multiple Choice
If carbon-12 had one more neutron, it would become...
boron-11
nitrogen-13
carbon-11
carbon-13
6
Multiple Choice
What is the name of Al2O3
aluminum oxide
dialuminum trioxide
aluminum III oxide
7
Multiple Choice
What is the name of the compound P4S10
phosphorous sulfide
tetraphosphide decasulfate
phosphorous decasulfate
tetraphosphorous decasulfide
8
Multiple Choice
What is the correct name for CF4
Carbon fluoride
Carbon IV fluoride
Carbon tetrafluoride
9
Multiple Choice
What is the correct name for the compound with the formula NH4NO3
ammonium nitrite
ammonium nitrate
ammonium nitride
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice
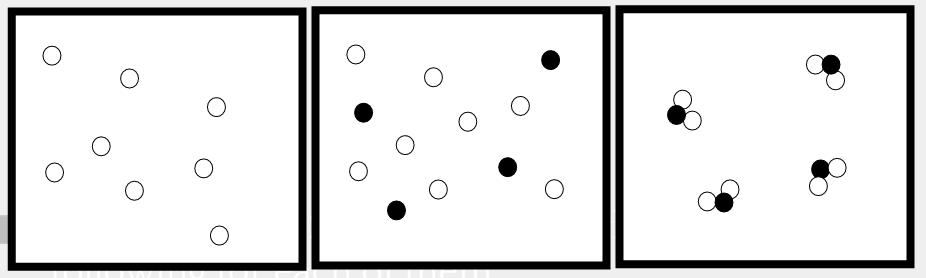
13
Multiple Choice

14
Multiple Choice

15
Multiple Choice
A combination that can be separated by physical processes is a .....
Pure Substance
Glucose
Element
Mixture
16
Multiple Choice
17
Multiple Choice
18
Multiple Choice

What is the number of protons that the element in this image contain?
14
7
15
18
19
Multiple Choice
An element has three isotopes. Given the abundances and relative masses, calculate the average atomic mass and determine (from the periodic table) which element it is.
Abundances | Relative masses
0.005% | 234.04 amu
0.720% | 235.04 amu
99.275% | 238.05 amu
Uranium (#92, Atomic Mass: 238.03 amu)
Fluorine (#9, Atomic Mass: 19.00 amu)
Mercury (#89, Atomic Mass: 200.59 amu)
Polonium (#84 209 amu)
20
Multiple Choice
21
Multiple Choice
How many neutrons are in an atom of carbon-14?
6
8
10
14
22
Multiple Choice
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
Na2S
Disodium sulfide
Disodium sulfide
Sodium sulfide
Sodium monosulfide
26
Multiple Choice
The correct way of showing the isotope of nitrogen with 7 protons and 8 neutrons is:
87N
78N
157N
715N
27
Multiple Choice
Atomic Number: 12
Mass Number: 25
Charge: 0
Most Common Isotope
Other Isotope
Cation
Anion
28
Multiple Choice
Neutrons: 42
Electrons: 36
Element: Arsenic
Most common isotope
Other Isotope
Anion
Cation
29
Multiple Choice
What is the most common isotope of boron?
boron-5
boron-10
boron-11
boron-13
30
Multiple Choice
What is the percent by mass of N in (NH4)2S?
47.1%
68.1%
44.1%
41.1%
Naming, Percent Composition, Matter, Atoms

Show answer
Auto Play
Slide 1 / 30
SLIDE
Similar Resources on Wayground

21 questions
Intro to mole
Presentation
•
10th Grade

24 questions
Lesson 6: The Development of a New Atomic Model
Presentation
•
10th Grade

26 questions
Nomenclature
Presentation
•
9th Grade

22 questions
Chemical Reactions Lesson for Lab
Presentation
•
10th Grade

22 questions
Cellular Respiration
Presentation
•
9th - 10th Grade

21 questions
CARBON(IV)OXIDE
Presentation
•
10th Grade

21 questions
Hybridisation and Molecular Geometry
Presentation
•
10th Grade

23 questions
Covalent Bonds Notes
Presentation
•
10th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade