

Electron Configruations
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
Standards-aligned
Benjamin Flo
Used 8+ times
FREE Resource
12 Slides • 15 Questions
1
Electron Configruations
Get Ready for new notes!

2
Multiple Choice
Each row on the periodic table represents:
an energy level
a sublevel
an electron
an orbital
3
Multiple Choice
Each row on the periodic table represents:
an energy level
a sublevel
an electron
an orbital
4
Multiple Choice
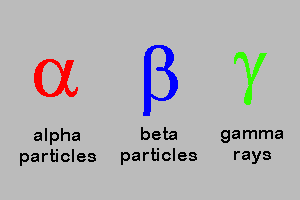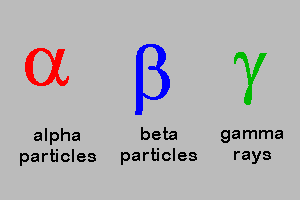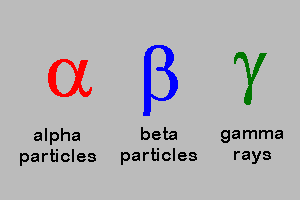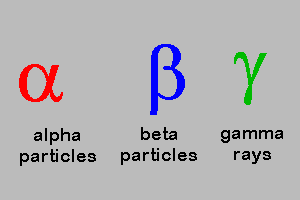
5
Open Ended
What's your good thing for today? What's your favorite TV show?
6

7

8

9

10

11

12

13

14

15

16
Please go to P-table.com
Press the "Raise Hand" button when you're ready to move on!
17
Multiple Choice

18
Multiple Choice
19
Multiple Choice

Based on the # of valence electrons, this could be the dot diagram of...
Mg
Cl
C
O
20
Multiple Choice
How many (valence) electrons should Flourine have around its Lewis dot model?
5
6
7
8
21
Multiple Choice
How many (valence) electrons should Beryllium have around its Lewis dot model?
1
2
3
4
22
Multiple Choice
Which area on the periodic table represents the electrons in the "d" sublevel?
representative elements
columns 3-8
rare earth elements
transition elements
23
Multiple Choice
1s22s22p63s2
24
Multiple Choice
25
Multiple Choice

26
Multiple Choice

27
Multiple Choice

Electron Configruations
Get Ready for new notes!

Show answer
Auto Play
Slide 1 / 27
SLIDE
Similar Resources on Wayground

19 questions
Reviewing Atoms, Ions, the Periodic Table, and Lewis dots
Presentation
•
10th - 12th Grade

21 questions
Ions
Presentation
•
8th - 11th Grade

21 questions
Cells Introduction
Presentation
•
9th - 12th Grade

18 questions
Valence Electrons
Presentation
•
9th - 12th Grade

22 questions
7.01: Electron Config, 7.02: Valence Electrons
Presentation
•
9th - 12th Grade

20 questions
Isotope and Atomic Mass
Presentation
•
9th - 12th Grade

20 questions
Atomic Numbers
Presentation
•
9th - 12th Grade

20 questions
Mass Number Atomic Number
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

15 questions
Nuclear Chemistry: Balancing
Quiz
•
10th Grade

20 questions
Naming Acids and Bases
Quiz
•
10th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade