

Electron Configuration- Practice
Presentation
•
Chemistry
•
10th Grade
•
Practice Problem
•
Easy
Standards-aligned
Manisha Midha
Used 487+ times
FREE Resource
14 Slides • 5 Questions
1
Electron Configuration Practice

2
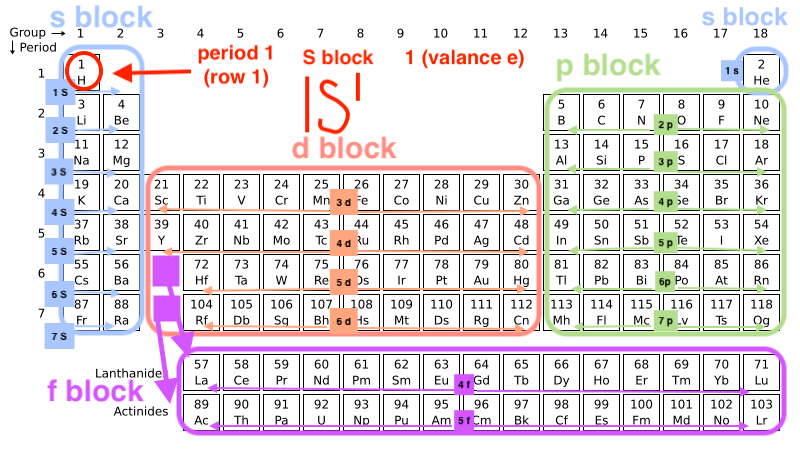
The Periodic Table
-Divided into different sections
-Helps to identify the energy levels of the elements

3
How to Write an Electron Configuration:
1. Start with the shell/period number
2. Followed by the type of orbital
3. Finally the superscript indicates the number of electrons in the orbital
Example: B = 1s2 2s2 2p1

4

5

6
Expanded Periodic Table
How the f block fits into the periodic table with the other energy levels
Read the energy levels from left to right, top to bottom

7

8
Hydrogen (1s1)
-The first element on the periodic table
-The element that starts the electron configuration levels
-1 electron on the inner most shell
9
Helium (1s2)
-The second element in period 1
-2 electrons on the inner most shell
-Start at H and move across the period to count the valence electrons

10
Beryllium (Be)
-The second element in period 2
-2 electrons on the second shell
-Start at H and move across the period to count the valence electrons
-Move down a period and count across, stopping at Be

11
Multiple Choice

Choose the correct electron configuration for Beryllium (Be).
1s1
1s2 2s2
1s2 2s2 2p1
12
Chlorine (Cl)
-The seventh element in period 3
-7 electrons on the third shell
-Start at H and move across the period to count the valence electrons
-Move down a period and count across
-Repeat, stopping at Cl

13
Multiple Choice

Choose the correct electron configuration for Chlorine (Cl).
1s2 2s2 2p1
1s2 2s2 2p6 3s2
1s2 2s2 2p6 3s2 3p5
14
Nickel (Ni)
-The tenth element in period 4
-8 electrons in 3s & 3p + 8 electrons in 3d block
-2 electrons on the fourth shell
-Start at H and move across the period to count the valence electrons
-Move down a period and count across
-Repeat, stopping at Ni

15
Multiple Choice

Choose the correct electron configuration for Nickel (Ni).
1s2 2s2 2p6 3s2 3p6 4s2 3d8
1s2 2s2 2p6 3s2 3p6 4s2 3d2
1s2 2s2 2p6 3s2 3p6 4s10
16
Krypton (Kr)
-The eighteenth element in period 4
-8 electrons in 3s & 3p + 8 electrons in 3d block
-8 electrons on the fourth shell (2 in 4s & 6 in 4p)
-Start at H and move across the period to count the valence electrons
-Move down a period and count across
-Repeat, stopping at Kr

17
Multiple Choice

Choose the correct electron configuration for Krypton (Kr).
1s2 2s2 2p6 3s2 3p6 4s8 3d8
1s2 2s2 2p6 3s2 3p6 4s2 3d8 4p3
1s2 2s2 2p6 3s2 3p6 4s2 3d8 4p6
18
Terbium (Tb)
-The eleventh element in period 6
-18 electrons in 4s, 4p, & 4d + 9 electrons in 4f block
-8 electrons in 5s & 5p
-2 electrons in the sixth shell
-Start at H and move across the period to count the valence electrons
-Move down a period and count across
-Repeat, stopping at Tb

19
Multiple Choice

Choose the correct electron configuration for Terbium (Tb).
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d19 5p6 6s2
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2 4f9
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2 4f5
Electron Configuration Practice

Show answer
Auto Play
Slide 1 / 19
SLIDE
Similar Resources on Wayground

17 questions
Nuclear radiations (part 1)
Presentation
•
10th Grade

13 questions
Exploring the Structure of Atoms
Presentation
•
10th Grade

16 questions
Ionic bonds
Presentation
•
10th Grade

13 questions
Periodic Trends
Presentation
•
10th - 11th Grade

16 questions
Solubility Curves, Water and Solutions
Presentation
•
10th Grade

16 questions
Chemical Bonding Ionic Covalent Metallic Lesson
Presentation
•
10th Grade

16 questions
Ionic Covalent and Metallic Bonding
Presentation
•
10th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

15 questions
Nuclear Chemistry: Balancing
Quiz
•
10th Grade

20 questions
Naming Acids and Bases
Quiz
•
10th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade