

Chemical Reaction
Presentation
•
Science
•
1st - 2nd Grade
•
Practice Problem
•
Medium
Standards-aligned
Rasheda Armstrong
Used 105+ times
FREE Resource
22 Slides • 13 Questions
1
Chemical Reaction
Ms. R. Armstrong

2
TYPES OF CHEMICAL REACTIONS
Combination
Decomposition
Precipitation
Neutralization
Combustion
Displacement
3
COMBINATION REACTION
Two or more compounds combine to form one compound.

4
DECOMPOSITION REACTION
The opposite of a combination reaction – a complex molecule breaks down to make simpler ones.

5
PRECIPITATION REACTION
Two solutions of soluble salts are mixed resulting in an insoluble solid (precipitate) forming.

6
NEUTRALISATION REACTION
An acid and a base react with each other. Generally, the product of this reaction is a salt and water.

7
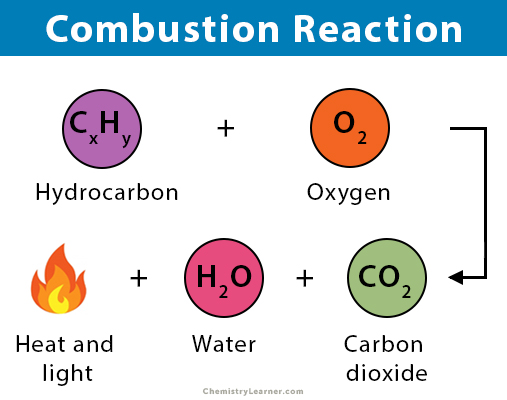
COMBUSTION REACTION
Oxygen combines with a compound to form carbon dioxide and water. These reactions are exothermic, meaning they give off heat
8
DISPLACEMENT REACTION
One element takes place with another element in the compound

9
Let's check what you remember
10
Multiple Choice
In order for neutralization to take place, a __________ and an ___________ must react with each other.
acid + salt
base + acid
water + salt
acid + water
11
Multiple Choice
When two compounds come together to form one compound, this is a
displacement reaction
combination reaction
precipitation reaction
combustion reaction
12
Multiple Choice
Combustion reactions give off heat
true
false
13
Multiple Choice
Which reaction does this represents?
A + BC → AC + B
displacement reaction
decomposition reaction
combustion reaction
combination reaction
14
Multiple Choice
Which reaction does this represent?
AB → A + B
Hint: It is the opposite of another reaction learnt
displacement reaction
combination reaction
decomposition reaction
combustion reaction
15
MIXTURES
A mixture is made from different substances that are not chemically joined.
NO CHEMICAL REACTIONS
16
TYPES OF MIXTURES
Homogenous
Heterogenous
17

18

19
HETEROGENOUS MIXTURE
A heterogeneous mixture is simply any mixture that is not uniform in composition.

20
...
Vegetable soup is a heterogeneous mixture. Any given spoonful of soup will contain varying amounts of the different vegetables and other components of the soup.

21
Skittles
This is a homogenous mixture
You can separate the pieces of candy physically

22
HOMOGENOUS MIXTURE
In this type of mixture, the components are evenly distributed throughout and the mixture has a uniform appearance

23
....
This orange juice was sweetened with some sugar.
Once the sugar particles have completely dissolved then it is considered a homogenous mixture

24
Multiple Choice

Is this a heterogenous mixture or homogenous mixture?
Heterogenous
Homogenous
25
Multiple Choice

What type of mixture is this?
homogenous
heterogenous
26
Multiple Choice

This is chocolate milk. What type of mixture is it?
homogenous
heterogenous
27
Multiple Choice

What type of mixture this sugar + water?
homogenous
heterogenous
28
WHAT IS A SOLUTION?
A solution is a homogeneous mixture of one or more solutes dissolved in a solvent.
29
SOLUTE
the substance that dissolves in a solvent to produce a homogeneous mixture
A substance that is dissolved in another substance (a solvent), forming a solution

30
SOLVENT
the substance in which a solute dissolves to produce a homogeneous mixture
A substance, ordinarily a liquid, in which other materials dissolve to form a solution.

31

32
Multiple Choice
Another name that can be used for homogenous mixture is
heterogenous
solvent
solute
solution
33
Fill in the Blanks
Type answer...
34
Fill in the Blanks
Type answer...
35
Poll
Are you ready far school next week?
Yes
A little
No
Chemical Reaction
Ms. R. Armstrong

Show answer
Auto Play
Slide 1 / 35
SLIDE
Similar Resources on Wayground

28 questions
ACTOS Y CONDICIONES
Presentation
•
1st Grade

32 questions
10.5 EDP Project Finale
Presentation
•
KG

25 questions
Patterns in the sky
Presentation
•
KG - 2nd Grade

29 questions
Scarcity
Presentation
•
2nd Grade

29 questions
Projecte de l’aigua
Presentation
•
1st - 3rd Grade

28 questions
3rd Recycle, Reduce, Reuse
Presentation
•
2nd Grade

30 questions
The Four Seasons
Presentation
•
KG

31 questions
Bądź uczciwy - przestrzeganie prawa w świecie informatyki
Presentation
•
1st Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

11 questions
Butterfly Life Cycle
Quiz
•
2nd Grade

14 questions
Frog Life Cycle
Quiz
•
2nd Grade

20 questions
EOY Science
Quiz
•
1st Grade

52 questions
Nature of Science and Investigations
Quiz
•
1st - 5th Grade

14 questions
Environmental Changes
Quiz
•
2nd Grade

10 questions
Identifying Physical and Chemical Changes
Interactive video
•
1st - 5th Grade

15 questions
habitats
Quiz
•
2nd Grade

14 questions
Parts of a Plant
Quiz
•
2nd Grade