

Chem Unit 1 Remediation- Density & Mass-Vol Graphs (LStd1.3,1.4)
Presentation
•
Chemistry, Physics, Science
•
9th - 12th Grade
•
Easy
Standards-aligned

John Wozniak (NCPS)
Used 8+ times
FREE Resource
25 Slides • 46 Questions
1
Chem - LStd 1.4 Remediation Lesson - Density

2

3
Multiple Choice
Which of the following is NOT a unit of density?
g/cm3
m3/g
g/ml
all are density units
4
Multiple Choice
Which of the following is NOT a unit of density?
kg/m3
m3/kg
kg/L
all are density units
5

6

7

8
Multiple Choice
9

10

11
Multiple Choice

12
Multiple Choice
the substances that are more dense ____________
will sink below the less dense substances
will float on the less dense substances
13
Multiple Choice
The substances that are less dense ______________
will float on the less dense substances
will sink below the less dense substances
14
Multiple Choice
A Balloon containing helium gas floats in the air because
helium gas is more dense than air
helium gas is less dense than air
15

16
Multiple Choice
The formula to calculate DENSITY (g/cm3) is
MASS x VOLUME
MASS - VOLUME
MASS / VOLUME
VOLUME/ MASS
17
Multiple Choice
18

19

20

21
Volume of the Blue Rectangular Prism
V = 10cm x 8cm x 9cm = 7200 cm3

22

23

24
Multiple Choice
The volume of irregular objects can be determined by using
water displacement method
water replacement method
water level method
25
Multiple Choice
26
Multiple Choice
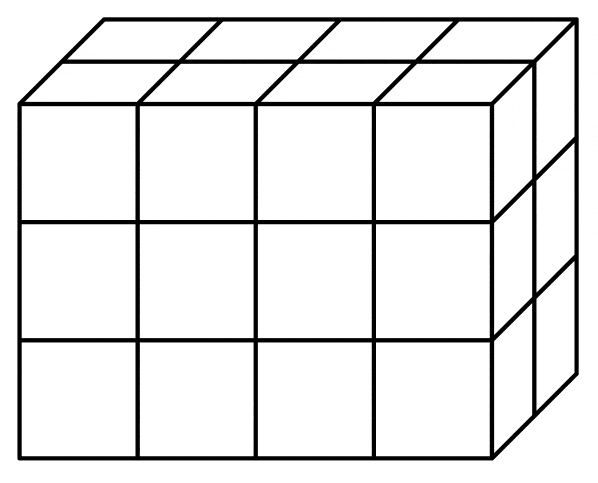
What is the volume of this rectangular prism?
26 cubic units
24 cubic units
24 square units
26 square units
27
Multiple Choice

Find the volume of the figure.
63 cm³
91 cm³
819 cm³
409.5 cm³
28
Multiple Choice

Each side of this ice cube measures 3 inches. What is the volume of the ice cube?
9 cubic inches (in3)
27 cubic inches (in3)
18 cubic inches (in3)
27 square inches (in2)
18 square inches (in2)
29
Multiple Choice

Each side of this ice cube measures 2 cm. What is the volume of the ice cube?
6 cm3
8 cm3
4 cm3
8 cm2
4 cm2
30
Multiple Choice
Steven finds the volume of a graduated cylinder with a penny in it. The volume was 43 mL. If the volume before the penny was added was 38 mL. Then what is the volume of the PENNY (=the displacement)?
43 mL
81 mL
38 mL
5 mL
31
Multiple Choice
Steven finds the volume of a graduated cylinder with a rock in it. The volume was 68 mL. If the volume before the rock was added was 41 mL. Then what is the volume displacement?
109 mL
68 mL
41 mL
27 mL
32

33
Multiple Choice
To calculate DENSITY, we must
take the amount of MASS in the object and DIVIDE it by the amount of VOLUME occupied by the object
take the amount of MASS in the object and MULTIPLY it by the amount of VOLUME occupied by the object
take the amount of MASS in the object and SUBTRACT it from the amount of VOLUME occupied by the object
take the amount of VOLUME occupied by the object and DIVIDE it by the amount of MASS in the object
34
Multiple Choice
Jack has a rock. The rock has a mass of 10 g and a volume of 2 cm3. What is the DENSITY of the rock?
5 mL
5 g/cm3
20 g/cm3
0.2 g/cm3
35
Multiple Choice
Jill also has a rock. Her rock has a mass of 2 g and a volume of 10 cm3. What is the DENSITY of the rock?
5 mL
5 g/cm3
20 g/cm3
0.2 g/cm3
36
Multiple Choice

Each side of this silver cube measures 2 cm. It has a mass of 84 g. What is the DENSITY of the silver cube?
42 g/cm3
10.5 g/cm3
21 g/cm3
8 square inches (in2)
37

38

39
0.5 g/cm3 x 800 cm3 = 400 g*cm3/cm3 = 400 g

40
Multiple Choice
When we solve the Density equation to find the Mass we see that MASS =
DENSITY x VOLUME
DENSITY / VOLUME
MASS / VOLUME
VOLUME / DENSITY
41
Multiple Choice
Jill has a rock. Her rock has a density of 2 g/cm3 and a volume of 10 cm3. What is the MASS of the rock?
20 g
20 g/cm3
5 g
0.2 g/cm3
0.2 g
42
Multiple Choice
Jack also has a rock. His rock has a density of 20 g/cm3 and a volume of 4 cm3. What is the MASS of the rock?
80 g
80 g/cm3
5 g
5 g/cm3
0.2 g
43
Multiple Choice

Each side of this silver cube measures 3 cm. It has a density of 10.5 g/cm3. What is the MASS of the silver cube?
31.5 g
94.5 g
283.5 g
0.29 g
0.028 g
44

45

46
(325 g) / (9.0 g/cm3) = 36.1 g/(g/cm3) = 36.1 g*(cm3/g) = 36.1 cm3

47
Multiple Choice
When we solve the Density equation to find the Volume we see that VOLUME =
DENSITY x MASS
DENSITY / MASS
MASS / VOLUME
MASS / DENSITY
48
Multiple Choice
Jill has a rock. Her rock has a density of 2 g/cm3 and a mass of 100 g. What is the VOLUME of the rock?
50 cm3
50 g/cm3
200 cm3
0.02 g/cm3
0.02 cm3
49
Multiple Choice
Jack also has a rock. His rock has a density of 10 g/cm3 and a mass of 20 g. What is the VOLUME of the rock?
200 cm3
2 g/cm3
2 cm3
0.5 g/cm3
0.5 cm3
50
Multiple Choice

This silver cube has a density of 10.5 g/cm3 and a mass of 210 g What is the VOLUME of the silver cube?
10.5 cm3
20 cm3
210 cm3
220.5 cm3
2205 cm3
51

52

53

54
Using a Mass Volume Graph to calculate the Density
"Making a Density Graph" video
Watch this short video
(copy the link and paste it into a new tab):
https://youtu.be/oVJPywo40Ps
55
Multiple Choice

For a Mass-vs-Volume graph, the slope of the line represents the _______________ of the substance.
volume
density
mass
length
56
Multiple Choice

Using this Mass-vs-Volume graph for Tin, calculate/find the density of Tin
10.0
7.3
4.8
3.0
57
Multiple Choice

Using this Mass-vs-Volume graph of Tin, what is the mass of a 1.20 cm3 sample?
0.20 g
1.20 g
8.75 g
10 g
58
Multiple Choice

Using this Mass-vs-Volume graph of Tin, what is the volume of a 5 g sample?
0.20 cm3
0.70 cm3
1.20 cm3
1.60 cm3
59
Multiple Choice

On a Mass-vs-Volume graph, the slope of the line represents the _______ of the substance
Density
Length
Mass
Volume
60
Multiple Choice

Using this Mass-vs-Volume graph, calculate/find the density of the substance
5.0 g/cm3
50.0 g/cm3
0.2 g/cm3
3.0 g/cm3
61
Multiple Choice

Using this Mass-vs-Volume graph, what is the volume of a 150 g sample?
10.0 cm3
20.0 cm3
30.0 cm3
40.0 cm3
62
Multiple Choice

Using this Mass-vs-Volume graph, what is the MASS of a 20.0 cm3 sample?
5 g
10 g
40 g
50 g
100 g
63
Multiple Choice

This is a Mass-vs-Volume graph of substances A, B, C, and D.
The slope of the line for Substance C represents the _______________ of Substance C.
volume
density
mass
length
64
Multiple Choice

Using this Mass-vs-Volume graph for Substances A, B, C, and D... Which substance has the highest density?
D
C
B
A
65
Multiple Choice

Using this Mass-vs-Volume graph for Substances A, B, C, and D... Which substance has the lowest density?
D
C
B
A
66
Multiple Choice

If you have EQUAL VOLUME samples of 4 different substances, which sample would have the largest MASS?
The sample with the HIGHEST DENSITY
The sample with the LOWEST DENSITY
67
Multiple Choice

Using this Mass-vs-Volume graph for Substances A, B, C, and D... If you have 10 cm3 samples of each substance, which sample would have the largest MASS?
D
C
B
A
68
Multiple Choice

If you have EQUAL MASS samples of 4 different substances, which sample would have the largest VOLUME?
The sample with the HIGHEST DENSITY
The sample with the LOWEST DENSITY
69
Multiple Choice

Using this Mass-vs-Volume graph for Substances A, B, C, and D... If you have 25g samples of each substance, which sample would have the largest VOLUME?
D
C
B
A
70
Multiple Choice
What is the MASS of a 10 cm3 sample of lead if lead has a density of 11.3 g/cm3?
0.88 g
1.13 g
113 g
1130 g
71
Multiple Choice
What is the VOLUME of a 10 g sample of lead if lead has a density of 11.3 g/cm3?
0.88 cm3
1.13 cm3
113 cm3
1130 cm3
Chem - LStd 1.4 Remediation Lesson - Density

Show answer
Auto Play
Slide 1 / 71
SLIDE
Similar Resources on Wayground

64 questions
Ch. 2 Analyzing Data
Presentation
•
9th - 12th Grade

68 questions
Rocks and The Rock Cycle
Presentation
•
9th - 12th Grade

65 questions
AP Biology FRQ Friday Tips and Tricks
Presentation
•
9th - 12th Grade

70 questions
Phylum Mollusca Notes
Presentation
•
10th - 12th Grade

69 questions
5.1.5 Life Science Practice
Presentation
•
9th - 12th Grade

65 questions
Unit 3.1 : Programming model - Register
Presentation
•
KG - University

67 questions
Unit 2: Chemistry Review
Presentation
•
9th Grade - University

64 questions
Proportional Parts in Triangles
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade