

Review and Moon
Presentation
•
Science
•
8th Grade
•
Medium
Rebecca Smith
Used 6+ times
FREE Resource
13 Slides • 52 Questions
1
Review and Moon

2
Part 1: Atoms
3
Atoms - Smallest Part of Matter
Electron: Positive, NUCLEUS
Neutron: No charge (neutral), NUCLEUS
Electron: Negative, ELECTRON CLOUD
4
Multiple Select
Which TWO of the following subatomic particles are inside the nucleus?
protons
electrons
neutrons
There are no subatomic particles in the nucleus of an atom.
5
Multiple Choice

Why is the overall charge of an atom neutral (0)?
They have the same number of protons and neutrons.
They have the same number of protons and electrons.
They have the same number of electrons and neutrons.
6
Multiple Choice

Which subatomic particle has a neutral (0) charge?
proton
nuetron
electron
atom
7
Multiple Choice

Which subatomic particle has a negative charge?
proton
neutron
electron
atom
8
Multiple Choice

Which subatomic particle has a positive charge?
proton
nuetron
electron
atom
9
Multiple Choice

The smallest particle into which an element can be divided and still be the same element is a(n)
proton
neutron
electron
atom
10
Atoms
Atomic Number = Number of Protons
Atomic Mass = Protons + Neutrons
Neutrons = Atomic Mass - Atomic Number
11
Fill in the Blanks

Type answer...
12
Fill in the Blanks

Type answer...
13
Multiple Choice

How many protons does Helium (He) have?
1
2
3
4
14
Multiple Choice

What does the 40.08 stand for in the Calcium block?
Atomic number
Atomic weight
Atomic symbol
15
Multiple Choice

What does the K stand for in the Potassium block?
Atomic number
Atomic weight
Symbol for the atom
16
Multiple Choice

What does the number 11 stand for in the Sodium block?
atomic number
atomic weight
Stranger Things
17
Multiple Choice

What is the chemical symbol of this element?
79
Au
AU
gold
18
Multiple Choice

What is the mass number of this element?
7
8
14
14.01
19
Multiple Choice
20
Multiple Choice
21
Multiple Choice
22
Atoms
Identity - Protons (ATOMIC NUMBER)
Reactivity - Valenvce Electrons
Outside energy level electrons
23
ATOMS - VALENCE ELECTRONS
Atoms WANT 8

24
Multiple Choice

How many valence electrons does Potassium Have?
1 Valence electron
2 Valence electron
3 Valence electron
4 Valence electron
5 Valence electron
25
Multiple Choice

How many valence electrons does Phosphorus Have?
1 Valence electron
2 Valence electron
3 Valence electron
4 Valence electron
5 Valence electron
26
Multiple Choice

How many valence electrons does Magnesium Have?
1 Valence electron
2 Valence electron
3 Valence electron
4 Valence electron
5 Valence electron
27
Multiple Choice

How many valence electrons does Helium Have?
1 Valence electron
2 Valence electron
3 Valence electron
4 Valence electron
5 Valence electron
28
Multiple Choice

How many valence electrons does Hydrogen Have?
1 Valence electron
2 Valence electron
3 Valence electron
4 Valence electron
5 Valence electron
29
Counting Atoms
Coefficient - BIG NUMBER IN THE FRONT - MULTIPLY THROUGH
Subscript - small number to the right of a SYMBOL or PARENTHESES - ONLY APPLIES TO THE THING TO THE LEFT OF IT.
30
Multiple Choice
How many atoms are there TOTAL in H2SO4 ?
6
5
7
3
31
Multiple Choice
32
Multiple Choice
How many Magnesium atoms are in 10MgCl2?
10
5
20
33
Multiple Choice
How many Oxygen atoms are in H2O?
1
2
0
4
34
Multiple Choice
How many Hydrogen atoms are in 4H2O?
6
8
2
4
35
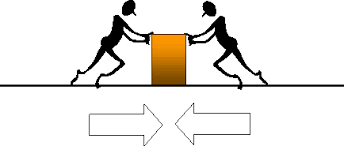
Forces
Balanced - an object not moving
Unbalanced - an object that is pushed or pulled
Force - push or pull
Force - Measured in NEWTONS
Net Force - total combined forces
Same direction - ADD
Opposite direction - SUBTRACT
36
Multiple Choice

Two forces act on an object to the right at the same time. What force is required to bring the object into equilibrium?
9 N to the right
9 N to the left
3 N to the left
18 N to the left
37
Multiple Choice

38
Multiple Choice
39
Multiple Choice
40
Multiple Choice

41
Multiple Choice
If the net force is equal to zero, the forces acting on the object are
balanced
unbalanced
42
Multiple Choice
43
Multiple Choice

44
Multiple Choice

45
Plate Boundary
Converge - COLLIDE
Diverge - DIVIDE
Transform - SLIDE

46
Multiple Choice

What type of boundary is this?
Divergent
Convergent
Transform
47
Multiple Choice

Which correctly identifies the geologic process that formed the Himalayan mountains (The World's Tallest Mountains, including Mt. Everest)?
Two continental plates converging
An earthquake in a subduction zone
A tectonic plate moving over a hot plate
Movement at a transform fault boundary
48
Multiple Choice

The Great Rift Valley in Africa and the Mid-Ocean Ridge were all created by this boundary
Divergent
Convergent
Reform
Transform
49
Multiple Choice

50
Phases of the Moon
29.5 day cycle
51
WAXING MOON - Increasing
Starts with New Moons
Ends with Full Moon
LIGHT ON THE RIGHT

52
WANING Moon - Decreasing
Full Moon back to New Moon
Light on the left.

53
Moon Phases
Each phases lasts about 3-5 days.
You can predict the phase of the moon.

54
Multiple Choice
55
Multiple Choice
56
Multiple Choice

57
Multiple Choice

58
Multiple Choice

59
Multiple Choice

60
Multiple Choice

61
Multiple Choice

62
Multiple Choice

63
Multiple Choice

64
Multiple Choice
65
Multiple Choice
Review and Moon

Show answer
Auto Play
Slide 1 / 65
SLIDE
Similar Resources on Wayground

63 questions
Newton's 2nd Law of Motion
Presentation
•
8th Grade

58 questions
Mitosis vs Meiosis
Presentation
•
7th Grade

61 questions
Adams & Jefferson Presidential Administrations
Presentation
•
8th Grade

55 questions
Earth Science Review Lesson
Presentation
•
8th Grade

55 questions
Science TCAP Practice
Presentation
•
8th Grade

61 questions
2nd 9 wk Review (Plate Tectonics, Lunar Cycle, HR, EMS)
Presentation
•
8th Grade

60 questions
Ecosystems & Cycles
Presentation
•
8th Grade

58 questions
Petroski - Physical Science LEAP Review
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade
Discover more resources for Science

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

46 questions
8th Science STAAR Review
Quiz
•
8th Grade

15 questions
Symbiotic Relationships
Quiz
•
6th - 8th Grade

10 questions
Food Chain and Food Web
Quiz
•
8th Grade

24 questions
8th grade Science STAAR Review
Quiz
•
8th Grade