

Isomers
Presentation
•
Chemistry
•
11th Grade
•
Practice Problem
•
Medium
Mr. Cavida
Used 37+ times
FREE Resource
13 Slides • 19 Questions
1
Isomers

2
Multiple Choice
Compounds that are composed of the same number and type of atoms but have them arranged in different ways are ___________.
isomers
isotopes
polymers
alkanes
3
Multiple Choice
There are two main forms of isomerism, which are?
Structural isomerism and Stereoisomerism
Stereoisomerism and Physical isomerism
Chemical isomerism and Stereoisomerism
Structural isomers and Geometric Isomers
4
Multiple Choice
In a stereoisomer, ________ means "on the same side" and ______ means "across from."
cis; trans
cis; L
D; L
trans; cis
5
Multiple Choice
6
Multiple Choice
A pair of molecules that exist in two forms that are mirror images of one another but cannot be superimposed.
Structural isomers
Sterioisomers
Geometric isomers
Enantiomers
7
Multiple Choice

Given the structural formulas:
Which two formulas represent compounds that are isomers of each other?
A and B
A and C
B and D
C and D
8
Multiple Choice

Given a formula representing a compound:Which formula represents an isomer of this compound?
9
Multiple Choice
Which formulas represent compounds that are isomers of each other?
10
What is isomerism?
Isomers are molecules with the same molecular formula (i.e. the same number and type of atoms) but in which the atoms are arranged in a different way.

11
Two main types of isomerism
Structural isomers have different structural formulae. Three types of structural isomerism are chain isomerism, positional isomerism and functional group isomerism.
Stereoisomers have the same structural formula, but the 3D arrangement of atoms is different (enantiomers). Two types are cis–trans isomerism and optical isomerism.

12
Chain isomerism
In chain isomers, the carbon chain is arranged differently.
For example, hexane has several chain isomers, all with the molecular formula C6H14:

13
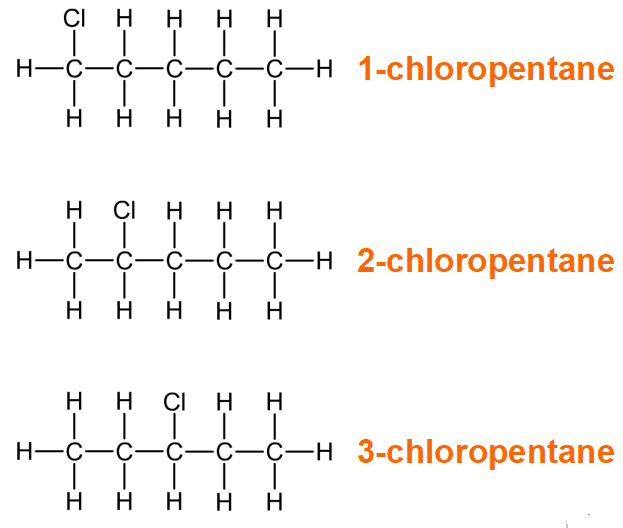
Positional isomerism
In positional isomers, the functional group is attached to a different carbon atom.
For example, chloropentane has several positional isomers, all with the molecular formula C5H11Cl:
14
Positional isomerism
Positional isomerism also exists in alkenes with four or more carbon atoms.
For example, hexene has several positional isomers, all with the molecular formula C6H12:

15
Functional group isomerism
Functional group isomers contain different functional groups and so are members of different homologous series.
For example, both alcohols and ethers have the general formula CnH2n+2O so they may be functional group isomers:

16
Multiple Choice

Chain isomers
Functional group isomers
Positional isomers
17
Multiple Choice

Chain isomers
Functional group isomers
Positional isomers
18
Multiple Choice

Chain isomers
Functional group isomers
Positional isomers
19
Multiple Choice

Chain isomers
Functional group isomers
Positional isomers
20
Multiple Choice

Chain isomers
Functional group isomers
Positional isomers
21
Cis–Trans isomerism
Molecules can rotate freely around single C-C covalent bonds, but not around C=C double bonds.
Isomers differ in the arrangement of the groups attached to the carbons in the double bonds.
These isomers cannot be superimposed on each other because the arrangement of the methyl groups is different.

22
Cis–Trans isomerism
If an alkyl group or atom other than hydrogen is attached to each carbon then the isomers can be named either cis (‘on the same side’) or trans (‘on the opposite side’).

23
Limitations of cis–trans isomerism
For example, is it possible to identify which of these halogenoalkanes is the cis isomer and which is the trans isomer?
Instead, a different system is used for these type of molecules: E–Z notation.

24
Limitations of cis–trans isomerism
In more complex organic compounds, in which multiple hydrogens have been substituted by different groups, isomers cannot be defined using the cis–trans notation.

25
E–Z isomerism
The E–Z notation is used to identify stereoisomers that cannot be called cis or trans.
Isomers are identified as either E or Z depending on what ‘priority’ is given to the groups attached to the carbon atoms in the double bond. The priority of these groups is determined by a complex series of rules.
E represents the German word ‘entgegen’, and corresponds to trans isomers. The highest priority groups are on the opposite side of the double bond.
Z represents the German word ‘zusammen’, and corresponds to cis isomers. The highest priority groups are on the same side of the double bond
26
Optical isomerism
A molecule can exist as two isomers that are non-superimposable, mirror images of each other, just like a left hand and right hand.
Optical isomers have the same physical properties, but they rotate polarized light in opposite directions.

27
Multiple Choice
Stereoisomers share the same structural formula.
True
False
28
Multiple Choice
Molecules cannot rotate about either single nor double covalent bonds.
True
False
29
Multiple Choice
Trans isomers mean the groups are at the same side of the double bond.
True
False
30
Multiple Choice
Trans isomers correspond to E isomers.
True
False
31
Multiple Choice
The prefixes cis- and trans- work for all types of stereoisomers.
True
False
32
Open Ended
How do you feel about this lesson? What are the topics that make you confuse at this moment?
Isomers

Show answer
Auto Play
Slide 1 / 32
SLIDE
Similar Resources on Wayground

23 questions
Elements and Compounds
Presentation
•
11th Grade

26 questions
Chemical Bonds
Presentation
•
10th Grade

26 questions
Types of chemical reactions
Presentation
•
10th Grade

23 questions
Naming Covalent Compounds
Presentation
•
10th Grade

23 questions
Stock System - Naming Ionic Compounds
Presentation
•
10th - 12th Grade

25 questions
Nutrients
Presentation
•
11th Grade

23 questions
Physics Chp 16 & 17 Doppler Effect & Speed of Sound with Videos
Presentation
•
11th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Redox Reactions
Quiz
•
10th - 12th Grade

20 questions
pH and pOH Calculations
Quiz
•
10th - 11th Grade

36 questions
Unit 4 (Part 2) Solutions
Quiz
•
10th - 12th Grade

30 questions
Final Exam Prep S22: 2.1.A.1 Isotopes
Quiz
•
9th - 12th Grade

25 questions
Stoichiometry Mass to Mass
Quiz
•
10th - 11th Grade

15 questions
Calculating pH and pOH
Quiz
•
10th - 12th Grade

15 questions
Balancing Chemical Equations
Quiz
•
10th - 12th Grade