

Chapter 10- Chemical Bonding
Presentation
•
Chemistry
•
University
•
Medium
+2
Standards-aligned

Luis Bello
Used 17+ times
FREE Resource
37 Slides • 33 Questions
1
Chapter 10- Chemical Bonding

2

3

4

5
Multiple Select
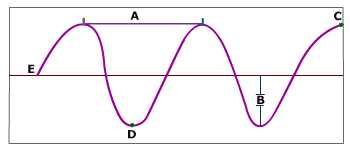
If the amplitude increases, the energy of the wave:
increases
decreases
stays the same
6
Multiple Choice

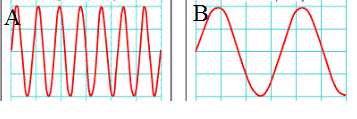
What is a wavelength?
How many waves pass a certain point?
The distance from one wave crest to another
The entire length of a wave
7
Multiple Choice
8
Multiple Choice
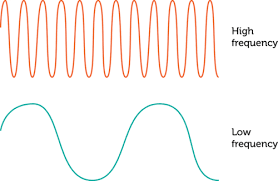
If a wave has a greater frequency, what happens to the wavelength?
increases
decreases
stays the same
9
Multiple Choice
10
Multiple Choice
11
Multiple Choice

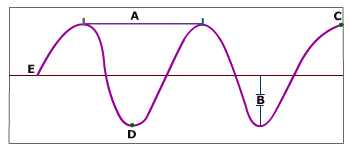
What does the arrow in the picture represent?
wavelength
echolocation
amplitude
peak
12
Multiple Choice
13
Multiple Choice
14
Multiple Choice

What is the correct set of quantum numbers for the last entering electron of magnesium?
n=2 l=0 ml=0 ms=-1/2
n=3 l=0 ml=0 ms=-1/2
n=3 l=1 ml=-1 ms=+1/2
n=3 l=0 ml=0 ms=+1/2
15
Multiple Choice

What orbital does the last entering electron of chlorine belong?
2s2
2p5
3p5
4d5
16
Multiple Choice

What is the correct electron configuration of silicon?
1s2 2s2 2p6 3p4
1s2 2s2 3p6 4s2 3d2
1s2 2s2 2p6 3s2 3p2
1s2 2s2 3p6 3s2 4p2
17
Multiple Choice

What type of sublevel has the this shape?
s
p
d
f
18

19

20

21

22

23
Multiple Choice

24
Multiple Choice
Which is the correct molecular structure for carbon dioxide?
25
Multiple Choice

How many total valence electrons are participating in bonding in the molecule above?
8
4
2
3
26
Multiple Choice
CO2 has how many lone pairs?
0
1
2
3
4
27
Multiple Choice
NH3 has how many lone pairs?
0
1
2
3
28

29

30

31

32

33

34
35

36
Second part...
By Luis Bello
37
38

39

40

41
Octet Rule
42

43
The octet rule states that atoms tend to form compounds in ways that give them eight valence electrons and thus the electron configuration of a noble gas.
An exception to an octet of electrons is in the case of the first noble gas, helium, which only has two valence electrons.
Atoms form compounds in ways that give them eight valence electrons.
Metals tend to lose electrons to achieve this configuration.
Nonmetals tend to gain electrons to achieve this configuration.
44
Cation Formation
Cations are the positive ions formed by the loss of one or more electrons. The most commonly formed cations of the representative elements are those that involve the loss of all of the valence electrons. Consider the alkali metal sodium (Na).
It has one valence electron in the third principal energy level. Upon losing that electron, the sodium ion now has an octet of electrons from the second principal energy level.
Cations form when an atom loses one or more electrons.
The resulting cation has the electron configuration of the noble gas atom in the row above it in the periodic table.
45
Anion Formation
Anions are the negative ions formed from the gain of one or more electrons. When nonmetal atoms gain electrons, they often do so until their outermost principal energy level achieves an octet. This process is illustrated below for the elements fluorine, oxygen, and nitrogen.
Anions are negative ions formed by accepting electrons.
The outermost principal energy level usually is an octet.
46

47
VSEPR Chart | Valence Shell Electron Pair Repulsion Theory
WHAT IS VSEPR THEORY?
The valence shell electron pair repulsion (VSEPR) theory is a model used to predict 3-D molecular geometry based on the number of valence shell electron bond pairs among the atoms in a molecule or ion.
This model assumes that electron pairs will arrange themselves to minimize repulsion effects from one another. In other words, the electron pairs are as far apart as possible.
48

49

50
VSEPR SHAPES
The VSEPR model is useful for predicting and visualizing molecular structures.
The structures are linear, trigonal planar, angled, tetrahedral, trigonal pyramidal, trigonal bipyramidal, disphenoidal (seesaw), t-shaped, octahedral, square pyramidal, square planar, and pentagonal bipyramidal.
51
52

53

54

55
Metallic Bond
Pure metals are crystalline solids, but unlike ionic compounds, every point in the crystal lattice is occupied by an identical atom.
The electrons in the outer energy levels of metal are mobile and capable of drifting from one metal atom to another.
This means that the metal is more properly viewed as an array of positive ions surrounded by a sea of mobile valence electrons.
Electrons that are capable of moving freely throughout the empty orbitals of the metallic crystal are called delocalized electrons.
A metallic bond is the attraction of the stationary metal cations to the surrounding mobile electrons.
56
Multiple Choice

57
Multiple Choice
58
Multiple Choice
59
Multiple Choice
60
Multiple Choice
61
Multiple Choice
62
Multiple Choice
63
Multiple Choice
64
Multiple Choice
65
Multiple Choice

66
Multiple Choice
67
Multiple Choice

The ------ model helps predict the shape of a molecule.
VESPR
Electron
Lewis
68
Multiple Choice
The ability of the metal to be drawn into wires.
Moving Electrons
Malleability
Ductility
69
Multiple Choice
What are charged particles that form when atoms gain or lose electrons?
Particle Atoms
Ions
Atomic Ions
Bonds
70
Multiple Choice
What do you call a bond that forms when electrons are transferred from one atom to another?
Compound Bond
Ionic Bond
Crystal Bond
Atom Bond
Chapter 10- Chemical Bonding

Show answer
Auto Play
Slide 1 / 70
SLIDE
Similar Resources on Wayground

64 questions
Bahasa Inggris 2 Meeting 6 and 7
Presentation
•
University

64 questions
Review on Sequence and Series
Presentation
•
KG - University

65 questions
Divergent Strabismus
Presentation
•
University

65 questions
PTA 110 Module 5 Pre Class Assignment
Presentation
•
University

64 questions
Chapter 1- The Chemical World
Presentation
•
University

62 questions
Propiedades Periódicas
Presentation
•
University

62 questions
ระบบคอมพิวเตอร์และประเภทคอมพิวเตอร์
Presentation
•
University

70 questions
Unit 1 level 3
Presentation
•
University
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

55 questions
Post Malone Addtion (Tres)
Quiz
•
12th Grade - University

20 questions
Disney Trivia
Quiz
•
University

50 questions
AP Biology Exam Review 2017
Quiz
•
11th Grade - University

24 questions
5th Grade Math EOG Review
Quiz
•
KG - University

12 questions
Star Wars Trivia - Easy
Quiz
•
KG - University

215 questions
8th Physical Science GA Milestones Review
Quiz
•
KG - University

40 questions
Famous Logos
Quiz
•
7th Grade - University

14 questions
(5-3) 710 Mean, Median, Mode & Range Quick Check
Quiz
•
6th Grade - University