

CA 4 Review
Presentation
•
Chemistry
•
12th Grade
•
Medium
+4
Standards-aligned
Simira Carothers
Used 10+ times
FREE Resource
6 Slides • 32 Questions
1
CA 4 Review
This Review will cover:
Acids and Bases
Thermochemistry

2
Acids and Bases
3
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
An Arrhenius acid:
donates H+ to another substance
accepts H+ from another substance
produces H+
produces OH-
7
Multiple Choice
An Arrhenius base:
donates H+
accepts H+
produces H+
produces OH-
8
Multiple Choice
A Bronsted Lowry acid:
donates H+ to another substance
accepts H+ from another substance
produces H+
produces OH-
9
Multiple Choice
A Bronsted Lowry base:
donates H+ to another substance
accepts H+ from another substance
produces H+
produces OH-
10
Multiple Choice
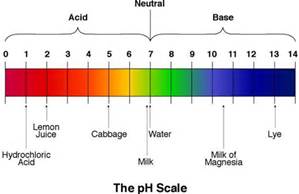
According to the pH range given in the image,which substance is more acidic than lemon juice?
Hydrochloric acid
Cabbage
Milk
Milk Of Magnesia
11
Multiple Choice
What type of reation occurs between an acid and a base?
Combustion
Synthesis
Neutralization
Double Replacement
12
Multiple Choice
Which of the following values would represent the pH of a strong base?
1
8
7
13
13
Multiple Choice
Which salt is formed in a reaction between HBr and LiOH?
LiBr
BrLi
NaCl
NaOH
14
Multiple Choice
Predict the products of this reaction.
HCl + Ca(OH)2 -->
NaCl
CaCl2
CaCl2 + H2O
H2O
15
Multiple Choice
What is the conjugate base in the following reaction?
HCO3- + HCl → H2CO3 + Cl-
HCO3-
HCl
H2CO3
Cl-
16
Multiple Choice
If the [H+] of a solution is 1.0 x 10-2 the pH is..
Hint: Use -log [H+]
17
Multiple Choice
What is the pH of a solution where the [H+] is 1.0 x 10-11?
11
13
14
1
18
Multiple Select
Which of the following is true about weak acid?
Hint: There are 2 answers
Acids that react partially with alkalis
Acids that are monobasic
Acid that produce low concentration of H+ ions
Acid that dissociates partially in H+ ions
19
Multiple Choice
Which of the following is the correct definition for a strong acid?
Partially dissociates into ions when dissolved in water and the change is not reversible
Fully dissociates into ions when dissolved in water and the change is not reversible
Fully dissociates into ions when dissolved in water and the change is reversible
Partially dissociates into ions when dissolved in water and the change is reversible
20
Thermochemistry
21
22
Multiple Choice
23
Multiple Choice
At higher temperature
particles in an object have less kinetic energy
particles in an object move faster
a gas contracts
particles in ab object move slower
24
Multiple Choice
The temperature of a substance increases as
the substance expands
the substance's volume increases
the substance's mass increases
the average kinetic energy of its particles increases
25
Multiple Choice

26
27
Multiple Choice

28
Multiple Choice

29
Multiple Choice

30
Multiple Choice

____ reactions usually feel cold.
endothermic
exothermic
31
Multiple Choice

A 500g piece of aluminum has a temperature of 7°C. What is the heat energy produced? Specific heat of Aluminum is 0.900 J/gºC.
3000 J
3000 g
3150 J
3150 g
32
Multiple Choice
33
Multiple Choice
34
Multiple Choice
35
Multiple Choice
36
Multiple Choice

How much heat does an aluminum block absorb if 10.00 grams is heated from 25.0oC to 50.0oC? *Specific heat of aluminum is 0.900 J/goC
450 J
-450 J
225 J
-225 J
37
Multiple Choice
38
Multiple Choice

The specific heat of platinum is 0.133 J/g°C. How much heat is released when a 10 g piece of platinum cools from 100°C to 50°C?
66.5 J
665 J
0.0266 J
0.665 J
CA 4 Review
This Review will cover:
Acids and Bases
Thermochemistry

Show answer
Auto Play
Slide 1 / 38
SLIDE
Similar Resources on Wayground

31 questions
Chap 5: Quantum Mechanical Model
Presentation
•
12th Grade

30 questions
Perhitungan Sifat Koligatif Larutan
Presentation
•
12th Grade

31 questions
PTS B.indonesia kelas 7
Presentation
•
12th Grade

29 questions
Lab Safety Is Common Sense
Presentation
•
KG

27 questions
Molecular Geometry
Presentation
•
11th - 12th Grade

30 questions
Lesson: nutrition, food preservation, body systems
Presentation
•
12th Grade

33 questions
% Yield, Limiting Reactants and Atom Economy
Presentation
•
11th - 12th Grade

34 questions
Materi Kompleksometri
Presentation
•
11th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade