

ATOMS Review
Presentation
•
Chemistry
•
8th Grade
•
Medium
Standards-aligned
Used 60+ times
FREE Resource
14 Slides • 11 Questions
1
ATOMS Review

2
Atoms make up all matter.

3
Protons IDENTIFY an element.

4
Neutrons have no charge, leaving the nucleus with always having a positive charge.

5
Electrons are in shells and determine reactivity.

6
Multiple Choice
7
Multiple Choice

The nucleus of an atom contains which subatomic particles?
protons and electrons
protons and neutrons
electrons and neutrons
protons, electrons, and neutrons
8
Multiple Choice
9
Multiple Choice

Which subatomic particle is counted to determine the type of element the atom represents?
proton
neutron
electron
boron
10

11

12

13
Multiple Choice
14
Multiple Choice

15

16
Multiple Choice

17
Multiple Choice

18
Multiple Choice

19

20

21

22

23

24
Multiple Choice
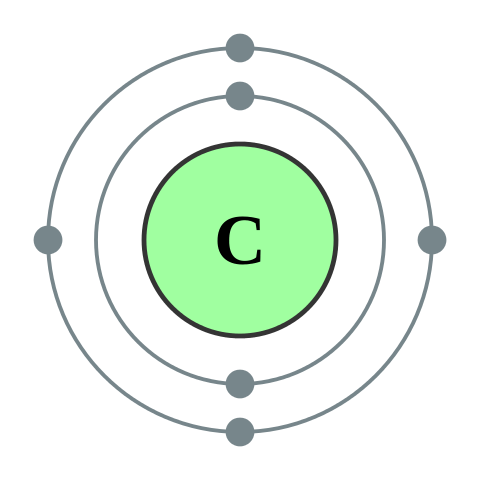
How many valence electrons does carbon have?
4
5
6
7
25
Multiple Choice

ATOMS Review

Show answer
Auto Play
Slide 1 / 25
SLIDE
Similar Resources on Wayground

20 questions
Parts of an Atom
Presentation
•
8th Grade

21 questions
Electrostatics
Presentation
•
8th Grade

21 questions
Electrical Conductors, Insulators, and Ways to Charge
Presentation
•
8th Grade

21 questions
FSSA Review - 8th Physical Science
Presentation
•
8th Grade

22 questions
States of Matter
Presentation
•
8th Grade

18 questions
Ionic Compounds
Presentation
•
8th Grade

20 questions
One and Two Step Equation Review
Presentation
•
9th Grade

21 questions
Atomic Structure
Presentation
•
8th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Acids and Bases
Quiz
•
8th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade

17 questions
Types of Chemical Reaction
Presentation
•
7th - 12th Grade

26 questions
Chem Inv 3
Quiz
•
6th - 8th Grade

20 questions
Counting Atoms
Quiz
•
8th Grade

33 questions
Acids and Bases Review
Quiz
•
6th - 8th Grade

41 questions
Atomic Structure and Periodic Table Unit Review
Quiz
•
8th Grade