

Parts of an Atom
Presentation
•
Science
•
8th Grade
•
Practice Problem
•
Medium
Standards-aligned
Laquinta Logan
Used 1K+ times
FREE Resource
11 Slides • 9 Questions
1
Parts of an Atom
2
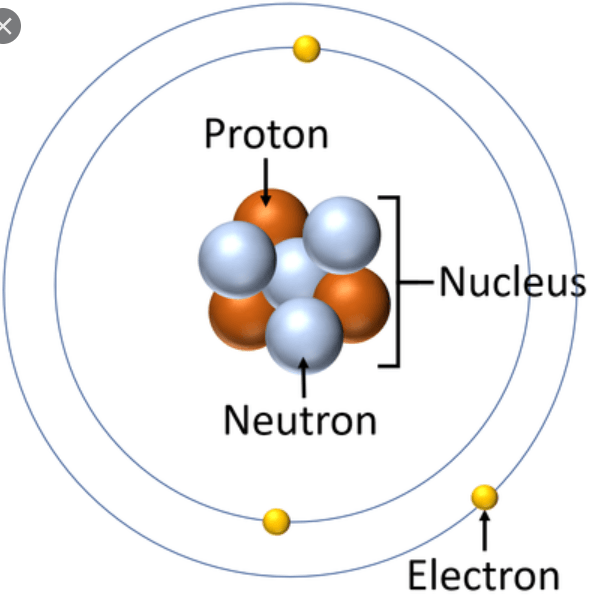
What is matter made of?
Matter is made from small particles called Atoms
Atoms are made from smaller particles called Subatomic Particles.
3 Subatomic particles are Protons, Neutrons, and Electrons
3
Multiple Select
What are the 3 Subatomic Particles?
(Choose all that apply)
nucleus
neutrons
protons
electrons
isotopes
4
Protons
Charge of a proton is Positive
Location of a proton is in Nucleus
Size of a proton is 1 AMU
5
Multiple Choice
Which statement is correct?
Protons are located outside the nucleus and have a positive charge.
Protons are located inside the nucleus and have a positive charge.
Protons are located outside the nucleus and have no charge.
6
Neutrons
Charge of the neutron is Neutral
(No Charge)
Location of the neutron is in the Nucleus
Size of the neutron is 1 AMU
7
Multiple Choice
Which statement is correct?
Neutrons are located outside the nucleus and have a positive charge.
Neutrons are located inside the nucleus and have a positive charge.
Neutrons are located inside the nucleus and have no charge.
8
Electrons
Charge of the electrons in Negative
Location of an electron is Outside the Nucleus
Size of an electron is so small that it adds no mass to the atom .
9
Multiple Select
Check all that are correct about an electron.
Electrons are positive
Electrons are negative
Electrons are inside the nucleus
Electrons are outside the nucleus
Electrons are very small
10
Nucleus
Nucleus contains protons and neutrons
Nucleus has a positive charge
Nucleus contains the mass of the atom
Nucleus is surrounded by the electron cloud
11
Multiple Choice
True or False: The nucleus contains the protons and electrons and has a positive charge.
True
False
12
Electron Cloud
Surrounds the nucleus
Contains electrons that are on energy shells
Has a negative charge
13
Multiple Choice
True or False: The electron cloud contains electrons and has a negative charge.
True
False
14
Energy Levels (Shells)
Energy levels/shells make up the electron cloud
Each energy level/shells holds a certain amount of electrons
We study the first 3 levels/shells
15
288 Rule
2 electrons on first energy level/shells
8 electrons on second energy level/shells
8 electrons on third energy level/shells
16
Multiple Select
Which is correct about the Energy Levels/Shells ? (select all that apply)
2 electrons on 1st energy level/shells
8 electrons on 2nd energy level/shells
8 electrons on 3rd energy level/shells
levels/shells holds electrons
17
Atomic Mass/ Mass Number
Protons and Neutrons contribute to the mass of the atom
Electrons DO NOT contribute to the mass of the atom BECAUSE they are TOO SMALL
PROTONS + NEUTRONS = Atomic Mass/ Mass Number
18
Fill in the Blanks
Type answer...
19
Neutral Atoms
Neutral atoms have to have the SAME number of protons and electrons because the (+) and (-) charges will cancel out.
20
Multiple Select
Neutral atoms must have the SAME number of which subatomic particles? (choose all that apply)
Protons
Neutrons
Electrons
Parts of an Atom

Show answer
Auto Play
Slide 1 / 20
SLIDE
Similar Resources on Wayground

17 questions
Plant & Animal Cells
Presentation
•
7th - 8th Grade

11 questions
Isotopes
Presentation
•
8th - 9th Grade

17 questions
Declaration of Independence
Presentation
•
8th Grade

12 questions
Prokaryote and Eukaryote Review
Presentation
•
9th Grade

12 questions
Phase Changes in Matter - Reading
Presentation
•
7th Grade

16 questions
Introduction to Cellular Respiration
Presentation
•
9th Grade

14 questions
Cell Structure and Funtions
Presentation
•
8th - 9th Grade

17 questions
Cell Theory & Levels of Organization
Presentation
•
7th - 9th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

45 questions
8th Grade Science - EOG Review
Quiz
•
8th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

10 questions
Exploring Earth's Seasons and Their Causes
Interactive video
•
6th - 8th Grade

10 questions
The Ocean's Role in Weather and Climate
Interactive video
•
6th - 8th Grade

10 questions
Evidence of Evolution
Presentation
•
6th - 8th Grade