

Electrolyte and Non Electrolyte
Presentation
•
Chemistry
•
12th Grade
•
Easy
Standards-aligned
Marissa sebastian Moe
Used 24+ times
FREE Resource
3 Slides • 10 Questions
1
Electrolyte and Non Electrolyte
Classifying substances into electrolytes and non-electrolytes

2
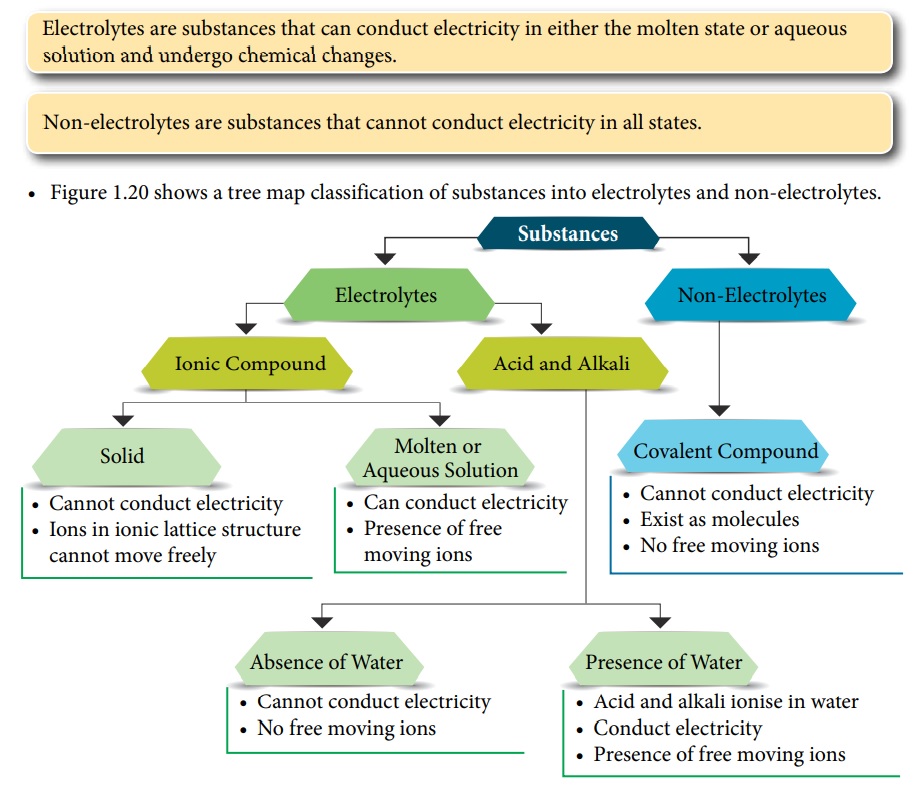
Analyze the following information before we proceed to the quiz section.
3
Multiple Choice
What is the the meaning of electrolyte?
Substances that can conduct electricity in either the molten state or aqueous solution and undergo chemical changes.
Substances that cannot conduct electricity in all states.
4
Multiple Choice
What is the the meaning of non-electrolyte?
Substances that can conduct electricity in either the molten state or aqueous solution and undergo chemical changes.
Substances that cannot conduct electricity in all states.
5
Multiple Choice
Which of the following is not an example of electrolyte?
Molten lead(II) bromide
Copper(II) chloride solution
Hydrochloric acid
Liquid ethanol
6
Multiple Choice
Why does covalent compound such as tetra chloromethane cannot conduct electricity in all state?
Exist as molecules and no free moving ions
Exist as solid
Has free moving ions
7
Multiple Choice
Ionic compounds are able to conduct electricity either molten or aqueous solution because
the presence of free moving ions
has no free moving ions
it is in molten or aqueous state
8
Multiple Choice
Ionic compounds are able to conduct electricity either molten or aqueous solution because
the presence of free moving ions
has no free moving ions
it is in molten or aqueous state
9
Multiple Choice
Acid and alkali can only conduct electricity
without the presence of water
with the presence of water
10
Multiple Choice
In the absence of water, acid and alkali cannot conduct electricity because
there is free moving ions
there is no free moving ions
11

The next questions will be based on this information. Analyze the table before you proceed.
12
Multiple Choice
What is the meaning of conductor?
Substances that conduct electricity in solid or molten state, but do not undergo chemical changes.
Substances that conduct electricity in molten state or aqueous solution, and undergo chemical changes.
13
Multiple Choice
Which of the following is an example of conductor?
Graphite
Acid and alkali
Electrolyte and Non Electrolyte
Classifying substances into electrolytes and non-electrolytes

Show answer
Auto Play
Slide 1 / 13
SLIDE
Similar Resources on Wayground

10 questions
Batteries Quizizz lesson
Presentation
•
KG

14 questions
Conduction, Convection, and Radiation in the Atmosphere
Presentation
•
KG - University

14 questions
Conduction Convection Radiation Worksheet
Presentation
•
KG - University

17 questions
Electrolysis of Molten Compounds
Presentation
•
12th Grade

6 questions
Solvation Vs. Dissociation
Presentation
•
9th - 12th Grade

15 questions
6.5 Concentration of Aqueous Solution
Presentation
•
10th Grade

16 questions
chem
Presentation
•
12th Grade

18 questions
Bonding - What it is and Why it happens
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

15 questions
Calculating pH and pOH
Quiz
•
10th - 12th Grade

20 questions
Le Chatelier's Principle
Quiz
•
9th - 12th Grade

40 questions
AP Chem Review: Units 6-9
Quiz
•
12th Grade

20 questions
Uranium: Twisting the Dragon's Tail
Quiz
•
10th - 12th Grade

13 questions
Nuclear Chemistry
Presentation
•
9th - 12th Grade

5 questions
DOL: Ideal Gas Law
Quiz
•
9th - 12th Grade

20 questions
pH/pOH Calculations
Quiz
•
12th Grade