

IB Bio Macromolecules Review
Presentation
•
Biology
•
11th - 12th Grade
•
Easy
+9
Standards-aligned
Jeffrey Nye
Used 8+ times
FREE Resource
49 Slides • 69 Questions
1
IB Bio Macromolecules Review
Covers Topics 2.1 - 2.5

2

3

4
Multiple Choice
sum of all chemical reactions
chemosynthesis
cellular respiration
metabolism
photosynthesis
5
Multiple Choice
The break down of larger molecules into small one.
metabolism
catabolism
anabolism
oxidation
6
Multiple Choice
Which is an anabolic process?
photosynthesis
cellular respiration
neither
both
7

8
Multiple Choice
The 4 most common elements in living things are:
carbon, hydrogen, oxygen, sulfur
carbon, hydrogen, oxygen, nitrogen
carbon, oxygen, sodium, calcium
carbon, oxygen, silicon, phosphorus
9

10

11
Multiple Choice

Which category does this molecule belong to?
carbs
lipids
proteins
nucleic acids
12
Multiple Choice

Which category does this molecule belong to?
carbs
lipids
proteins
nucleic acids
13
Multiple Choice

Which category does this molecule belong to?
carbs
lipids
proteins
nucleic acids
14
Multiple Choice

Which category does this molecule belong to?
carbs
lipids
proteins
nucleic acids
15
Multiple Choice

Which category does this molecule belong to?
carbs
lipids
proteins
nucleic acids
16
Multiple Choice

Which category does this molecule belong to?
carbs
lipids
proteins
nucleic acids
17

18

19
Multiple Choice
The oxygen atom in a water molecule has a partial ______ charge.
negative
positive
20
Multiple Choice
Water is made up of two hydrogen atoms joined by a _______ bond to an oxygen atom.
covalent
ionic
hydrogen
21
Multiple Choice

In the water molecule shown, which atoms should be labeled with a partial positive charge?
hydrogens
oxygen
both
neither
22

23
Multiple Choice
Water forms ____________ between water molecules.
aquatic bonds
covalent bonds
hydrogen bonds
ionic bonds
24

25
Multiple Choice
Water sticking to water is _________ ; water sticking to other charged substances is __________.
magic; science
adhesion; cohesion
cohesion; adhesion
cohesion; capillary action
26
Multiple Choice
Water molecules are _______ and can dissolve other _______ substances.
nonpolar; nonpolar
polar; nonpolar
nonpolar; polar
polar; polar
27

28
Multiple Choice
Which best describes the thermal properties of water?
Because of hydrogen bonding, water must absorb a large amount of energy to change temperature or state
Because of hydrogen bonding, water must absorb a small amount of energy to change temperature or state.
Water's temperature is unstable and changes easily with its surroundings.
Water and methane are similar in that they are both liquid at room temperature.
29

30
Multiple Choice
Which of the following is insoluble in water?
glucose
oxygen
sodium chloride
fatty acids
31
Multiple Choice

Which property of water is shown here?
cohesion
adhesion
thermal
solvent
32
Multiple Select
Hydrogen bonding is responsible for which properties of water? (select all that apply)
cohesive properties
adhesive properties
thermal properties
solvent properties
33

34

35
Multiple Choice
(Why do I need this before a workout?)
36

37

38
Multiple Choice
Which of the following is NOT a monosaccharide?
Glucose
Galactose
Fructose
Lactose
39

40

41

42
Multiple Choice

43
Multiple Select

Which of the following are disaccharides?
Sucrose
Glucose
Maltose
Cellulose
44

45
Multiple Choice
This carbohydrate is used by animals to store energy in the liver.
chitin
glycogen
glucose
starch
46
Multiple Choice
Energy storage in plants is in ______ molecules.
cellulose
starch
glycogen
47

48
Multiple Choice
49
Multiple Choice

50

51
Multiple Select
Which of the following is a function of lipids? Select all that apply
Long-term energy storage
Short-term energy storage
Insulation
Waterproof coverings
52
Multiple Choice
Lipids are... (Hint: this is due to the hydroPHOBIC nature of the phospholipid tails)
Water Fearing
Hight Fearing
Chicken Fearing
Scratch Fearing
53

54
Multiple Choice
In addition to glycerol, how many fatty acids are needed to make a triglyceride?
3
4
2
1
55
Multiple Choice

Which biomolecule is this?
carbohydrate
triglyceride
phospholipid
steroid
56

57
Multiple Choice
This macromolecule is used for long term energy storage.
protein
simple carbohydrate
complex carbohydrate
lipid
58

59
Multiple Choice
More than one double bond
Monounsaturated fatty acid
Unsaturated fatty acid
Polyunsaturated fatty acid
Polysaccharides
60
Multiple Choice
All single bonds between carbons
cis or trans isomers
Monounsaturated fatty acid
Polyunsaturated fatty acid
Saturated fatty acid
61
Multiple Choice
Which is an unsaturated fatty acid?
62
Multiple Choice

This molecule is...
Saturated
Unsaturated
63

64
Multiple Choice
Hydrogen atoms at a double bond are on the same side; bends; oils
hydrolysis
Cis
Trans
Glycogen
65
Multiple Choice

What type of fatty acid is shown here?
saturated fatty acid
cis-unsaturated fatty acid
trans-unsaturated fatty acid
cis-polyunsaturated fatty acid
66
Multiple Choice

This is an example of ______-Oleic acid.
trans
cis
67

68

69
Multiple Choice
Unsaturated fats are bad for you
True
False
70

71

72
Multiple Choice
What is a protein:
A polymer composed of many sugars.
A polymer composed of many amino acids.
A polymer composed of fatty acids.
A polymer composed of nucleotides.
73

74
Multiple Choice
75

76
Multiple Choice
77
Multiple Choice
Which shows a peptide bond?
78
Multiple Choice
79
Multiple Choice

The diagram shows a bond forming between two amino acids. What is the name of this reaction?
Condensation Synthesis
Hydrolysis
Pepysis
Oxidation
80

81
Multiple Choice
This is one job proteins do NOT have in the body
storing genetic information
structure
speed up chemical reactions
transport things through cell membrane
82
Multiple Choice
The enzyme that catalyses the first step of carbon fixation in photosynthesis
spider silk
Rhodopsin
Rubisco
Proteome
83
Multiple Choice
A structural protein that forms strong fibers, found in skin, connective tissue and bone (the most abundant protein in the animal kingdom).
Insulin
Immunoglobulins
Rhodopsin
Collagen
84
Multiple Choice
A 4 polypeptide globular protein in red blood cells. Binds oxygen in lungs and releases it in tissues.
Integrin
Hemoglobin
Lysozyme
Rubisco
85

86
Multiple Choice
87

88

89
Multiple Choice

90

91
Multiple Choice
92

93
Multiple Choice

94

95
Multiple Choice
96
Multiple Choice
97
Multiple Choice
98

99
Multiple Choice
100

101
Multiple Choice
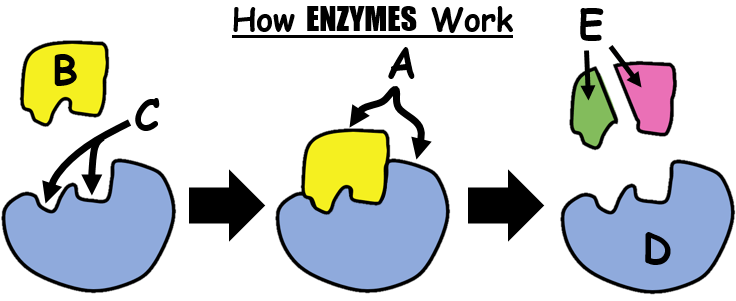
Letter C...
active site
enzymes
substrate
products
102
Multiple Choice
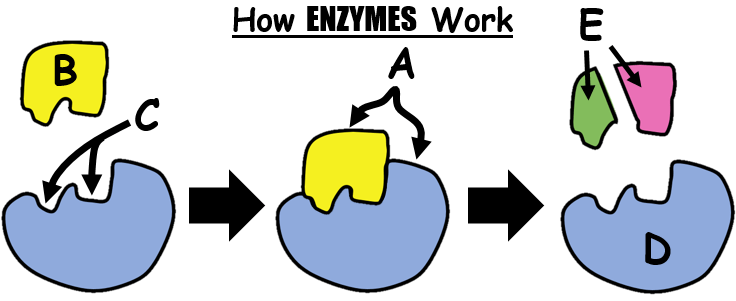
103

104

105
Multiple Choice
106

107
Multiple Choice
What happens when enzymes are heated to a high temperature?
The enzymes die.
The shape is altered and they denature
The enzymes' amino acid sequence changes.
The enzymes remain the same
108

109
Multiple Choice
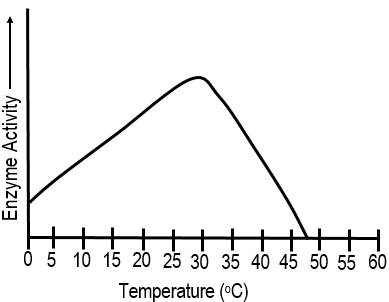
110

111
Multiple Choice
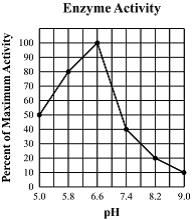
112
Multiple Choice
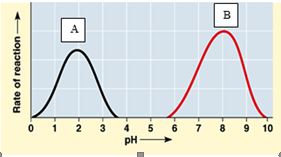
113
Multiple Choice
114

115

116
Multiple Choice
What is the advantage of using lactase in an immobilized state in the food manufacturing industry?
It functions within cells.
It dissolves in multiple solvents.
It converts cellulose into glucose.
It is less likely to become denatured.
117

118
Multiple Choice
What is decreased when lactase is added to milk?
A. Sweetness
Disaccharides
Calcium
Monosaccharides
IB Bio Macromolecules Review
Covers Topics 2.1 - 2.5

Show answer
Auto Play
Slide 1 / 118
SLIDE
Similar Resources on Wayground

112 questions
Interdependence of Organisms in Their Environment
Presentation
•
9th - 12th Grade

118 questions
Bisnis Digital
Presentation
•
12th Grade

118 questions
Vitamins Lecture
Presentation
•
University

115 questions
WW1 USH
Presentation
•
9th - 12th Grade

113 questions
The Civil Rights Movement
Presentation
•
11th Grade

118 questions
IB Biology Carbohydrates
Presentation
•
11th - 12th Grade

115 questions
DC US History
Presentation
•
12th Grade

112 questions
Industrialization Part 2
Presentation
•
KG - University
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Biology

50 questions
NC EOC Biology Review part 1
Quiz
•
9th - 12th Grade

40 questions
Biology EOC Review
Quiz
•
11th Grade

17 questions
Communicable and Chronic Diseases Slidedeck
Presentation
•
9th - 12th Grade

140 questions
Biology Final Quizizz
Quiz
•
10th - 11th Grade

20 questions
NC EOC Biology Review part 1
Quiz
•
9th - 12th Grade

64 questions
Disease Unit- 2026
Quiz
•
9th - 12th Grade

22 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
9th - 11th Grade

55 questions
Mock EOC/ Interim 3 Review
Quiz
•
KG - University