

Carbon
Presentation
•
Biology
•
7th Grade
•
Medium
Jessave Arbias
Used 3+ times
FREE Resource
25 Slides • 34 Questions
1
Review Part 1by T Bry

2
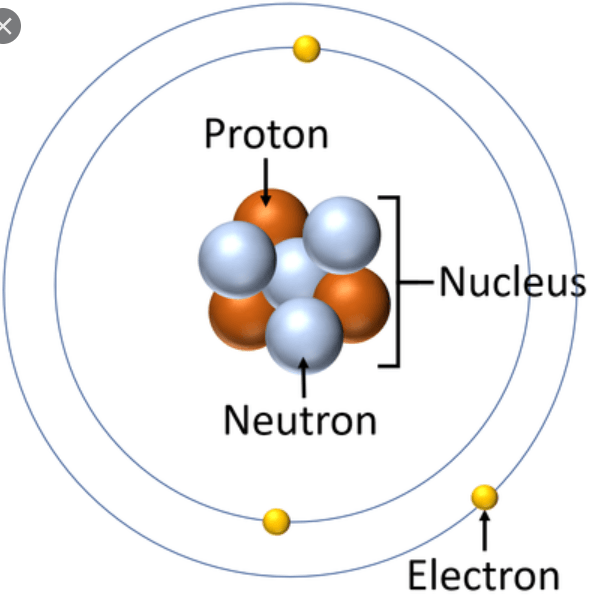
Parts of an Atom
3
Multiple Choice

4
Multiple Choice
5
Multiple Choice

6
Multiple Choice

7
Multiple Choice

8
Multiple Choice

9
Multiple Choice

10
Multiple Choice

11
What is matter made of?
Matter is made from small particles called Atoms
Atoms are made from smaller particles called Subatomic Particles.
3 Subatomic particles are Protons, Neutrons, and Electrons
12
Multiple Select
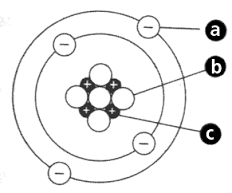
What are the 3 Subatomic Particles?
(Choose all that apply)
nucleus
neutrons
protons
electrons
isotopes
13
Protons
Charge of a proton is Positive
Location of a proton is in Nucleus
Size of a proton is 1 AMU
14
Multiple Choice
Which statement is correct?
Protons are located outside the nucleus and have a positive charge.
Protons are located inside the nucleus and have a positive charge.
Protons are located outside the nucleus and have no charge.
15
Neutrons
Charge of the neutron is Neutral
(No Charge)
Location of the neutron is in the Nucleus
Size of the neutron is 1 AMU
16
Multiple Choice
Which statement is correct?
Neutrons are located outside the nucleus and have a positive charge.
Neutrons are located inside the nucleus and have a positive charge.
Neutrons are located inside the nucleus and have no charge.
17
Electrons
Charge of the electrons in Negative
Location of an electron is Outside the Nucleus
Size of an electron is so small that it adds no mass to the atom .
18
Multiple Select
Check all that are correct about an electron.
Electrons are positive
Electrons are negative
Electrons are inside the nucleus
Electrons are outside the nucleus
Electrons are very small
19
Nucleus
Nucleus contains protons and neutrons
Nucleus has a positive charge
Nucleus contains the mass of the atom
Nucleus is surrounded by the electron cloud
20
Electron Cloud
Surrounds the nucleus
Contains electrons that are on energy shells
Has a negative charge
21
Multiple Choice
True or False: The electron cloud contains electrons and has a negative charge.
True
False
22
Energy Levels (Shells)
Energy levels/shells make up the electron cloud
Each energy level/shells holds a certain amount of electrons
We study the first 3 levels/shells
23
288 Rule
2 electrons on first energy level/shells
8 electrons on second energy level/shells
8 electrons on third energy level/shells
24
Atomic Mass/ Mass Number
Protons and Neutrons contribute to the mass of the atom
Electrons DO NOT contribute to the mass of the atom BECAUSE they are TOO SMALL
PROTONS + NEUTRONS = Atomic Mass/ Mass Number
25
Fill in the Blanks
Type answer...
26
Neutral Atoms
Neutral atoms have to have the SAME number of protons and electrons because the (+) and (-) charges will cancel out.
27

28

29

30

31
Multiple Choice
Which substance is not an element?
Neon (Ne)
Oxygen (O2)
Carbon dioxide (CO2)
Iron (Fe)
32

33
Multiple Choice
Which substance is a compound?
Neon (Ne)
Oxygen (O2)
Carbon Dioxide (CO2)
Nitrogen (N2)
34

35
Multiple Choice
Which substance is not a molecule?
Neon (Ne)
Oxygen (O2)
Carbon Dioxide (CO2)
Nitrogen (N2)
36
Multiple Choice
Is Sodium (Na) an element or a compound?
Element
Compound
37
Multiple Choice
Is Sodium Chloride (NaCl) an element or compound?
Element
Compound
38
Multiple Choice
Is Iron (Fe) an atom or a molecule?
Atom
Molecule
39
Multiple Choice
Is Chlorine (Cl2) an atom or a molecule?
Atom
Molecule
40
Multiple Select
Neutral atoms must have the SAME number of which subatomic particles? (choose all that apply)
Protons
Neutrons
Electrons
41

42
Multiple Select
Select all that are made up of molecules.
sandwiches
atoms
people
chair
43

44
Multiple Choice
Where do we get glucose molecules from?
Sugars/Starches
Protein
Air
Water
45
Multiple Choice
Which organic compound functions MAINLY to provide short-term energy storage for your body?
Carbohydrates
Lipids
Proteins (Polypeptides)
Nucleic Acids
46

47
Multiple Select
Select all that contain protein.
meat
eggs
beans
candy
48
Multiple Choice
Which organic compound provides long-term energy storage for your body?
Carbohydrates
Lipids
Proteins (Polypeptides)
Nucleic Acids
49
Multiple Choice
Which organic compound stores and communicates information in your cells?
Carbohydrates
Lipids
Proteins (Polypeptides)
Nucleic Acids
50
Nucleic Acids
DNA and RNA molecules encode the information needed to build and maintain your body.

51
Proteins
Proteins (polypeptides) are made from building blocks called amino acids. There are 22 types of amino acids! Eating a wide variety of foods is important to make sure your body is getting all essential amino acids.

52
Multiple Choice
Last but not least... which of the following is NOT a main function of proteins in your body?
Structural support
Membrane transport
Signalling
Energy
53
Multiple Select
Digesting starch (a polysaccharide) into maltose (a disaccharide) subunits would be described as... (check all that apply)
Anabolism
Catabolism
Condensation
Hydrolysis
54
Multiple Select
Assembling proteins from amino acids (monomers) would be described as... (check all that apply)
Anabolism
Catabolism
Condensation
Hydrolysis
55
Metabolism = Anabolism + Catabolism
Catabolic reactions break complex molecules apart to make simple molecules
Catabolism relies on hydrolysis reactions

56
Metabolism = Anabolism + Catabolism
Anabolic reactions add simple molecules together to make complex molecules
Anabolism relies on condensation reactions

57
Multiple Choice
"-saccharide" is a suffix that means "sugar." Which image below shows a monosaccharide?
58
Multiple Choice
Which image below shows a polysaccharide?
59
Multiple Choice
Which image below shows a polysaccharide?
Review Part 1by T Bry

Show answer
Auto Play
Slide 1 / 59
SLIDE
Similar Resources on Wayground

57 questions
Periodic Table Quiz Lesson Review
Presentation
•
7th Grade

54 questions
Human Impact on Natural Resources
Presentation
•
7th Grade

54 questions
Friction and Net Force
Presentation
•
7th Grade

55 questions
Weather in the Atmosphere
Presentation
•
6th Grade

56 questions
Simple and Extended Structures
Presentation
•
7th Grade

52 questions
LEAP REVIEW #1
Presentation
•
6th Grade

53 questions
Weathering, erosion and deposition
Presentation
•
7th Grade

52 questions
Solar System Exploration Review Honors 22-23
Presentation
•
6th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Biology

20 questions
Food Chains and Food Webs
Quiz
•
7th - 12th Grade

10 questions
Exploring Evidence of Evolution
Interactive video
•
6th - 10th Grade

20 questions
7.13 A PC: Skeletal/Muscular/Circulatory/Respiratory
Quiz
•
7th Grade

10 questions
Energy Flow and Matter Recycling in Ecosystems
Interactive video
•
6th - 10th Grade

20 questions
Ecology quiz updated (2026)
Quiz
•
7th Grade

10 questions
The Nitrogen Cycle
Quiz
•
6th - 10th Grade

16 questions
Evidence of Evolution
Quiz
•
6th - 7th Grade

17 questions
6 Kingdoms of Life
Quiz
•
6th - 8th Grade