

Separation Techniques
Presentation
•
Chemistry, Science
•
7th - 10th Grade
•
Medium
X Y
Used 14+ times
FREE Resource
29 Slides • 24 Questions
1
Separation Techniques

2
Learning Objective
At the end of the lesson you should able to:
understand the difference between pure substance and mixtures.
Some text here about the topic of discussion
3
A mixture is made up of two or more substances that are not chemically combined together.
Mixtures
Mixtures
Some text here about the topic of discussion

4

When two or more elements physically combined. Alloy is an example of a mixture of elements .
Some text here about the topic of discussion.
Mixtures of elements
5

When two or more compounds physically combined. Seawater is an example where salt and water physically combined.
Some text here about the topic of discussion.
Mixture of compounds
6

When two or more compounds and elements physically combined. Surround air is a very good example.
Some text here about the topic of discussion.
Mixture of compounds and elements
7
Open Ended
Name a mixture that you encounter in your everyday life.
8
Pure Substance
A pure substance is made up of one single element or compound. It is not mixed with any other substance.
Fun fact:
Diamond is highly compressed carbon element
Some text here about the topic of discussion

9
10
Examples of mixtures:
Filtration is used to separate insoluble particles from a liquid.
Filtration
Some text here about the topic of discussion
sand and water
chalk powder and water (suspension)
•clay and water

11
Open Ended

What do you think the picture is trying to illustrate about filtration?
12
Multiple Choice
During filtration, the solid that is trapped in the filter paper is called ..A.. and the liquid that has passed through the filter paper is called ..B..
A: residue B: filtrate
A: residue B: filterate
A: filtrate B: residue
A: filterate B: residue
13

This is a process of obtaining a soluble solid from a solution by heating the solution until all the water has boiled off.
Some text here about the topic of discussion.
Evaporation to Dryness
14

Substances that decompose on heating cannot be obtained by evaporation to dryness.
e.g. Sugar decomposes on heating
Some text here about the topic of discussion.
Evaporation to Dryness
15
Process of obtaining pure solid sample (soluble solid) from its solution.
Some text here about the topic of discussion.
Crystallisation


Copper (II) Sulfate Crystals
16
Evaporation to dryness and Crystallisation
https://www.youtube.com/watch?v=vi_SJBnxmHo&ab_channel=Cognito
17
Multiple Choice
Which separation technique is used to obtain salt from seawater?
Simple distillation
Filtration
Chromatography
Evaporation
18
Fill in the Blanks
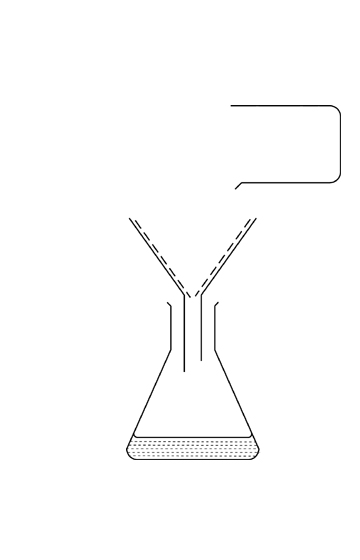
Type answer...
19
Simple Distillation
Simple distillation is used to separate a pure solvent (liquid) from a solution.
Examples:
Extraction of water from:
•sea water
•salt (sodium chloride) solution
•sugar solution
Some text here about the topic of discussion
20

21







22
Multiple Choice
Which separation technique is used to obtain pure water from seawater?
Simple distillation
Filtration
Chromatography
Evaporation
23
Multiple Choice
Which of the following statements tells us that a sample of liquid is pure water?
It is colourless.
It leaves no residue when filtered.
It boils at 100oC.
It is odourless.
24
Multiple Choice
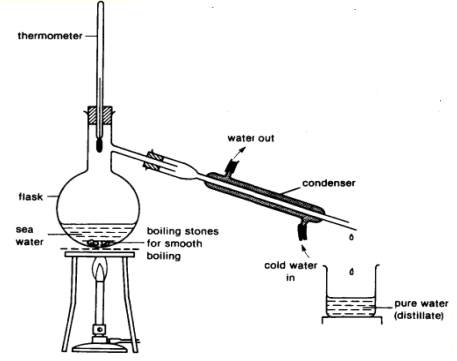
What is the name of this separation technique?
Simple distillation
Fractional distillation
Boiling
Evaporation
25

This method can be used to separate a mixture of miscible liquids with different boiling points.
Liquids that mix together completely to form a solution are described as miscible.
Examples: Ethanol and water
Some text here about the topic of discussion.
Fractional Distillation
26





27




28

Recall:
From Chapter 1 Kinetic Particle Theory:
what physical states were present when the temperature is constant?
Why did the temperature remain constant?
Some text here about the topic of discussion.
Heating curve of Fractional Distillation
29

To separate:
the various components of crude oil such as petrol, kerosene, diesel in oil refineries.
nitrogen, oxygen and argon in liquid air.
ethanol from the fermentation mixture of water, glucose and malt in breweries.
Some text here about the topic of discussion.
Industrial Application of Fractional Distillation
30
Fractional Distillation of Air
31
Multiple Choice
What is the physical property on which fractional distillation depends?
different boiling points
different melting points
different solubilities
different densities
32
Chromatography is the method of separating two or more components that dissolve in the same solvent.
Some text here about the topic of discussion.
Paper Chromatography



33
Open Ended
Can you think of any use of colour pigment in our daily live?
34
35
Depend on relative solubility of the component
More soluble travel faster and further from start line
less soluble travel slower and closer to start line
Which color pigment in the picture is the most and least soluble?
Principle behind paper chromatography

36
Identical dyes travel up the same distance and produce the same colour on the paper when the same solvent is used
The chromatography paper with the separated components is called a chromatogram.
Which color pigment in the picture is the most and least soluble?
Principle behind paper chromatography

37
Open Ended

Why is the chromatogram on the right is a pure substance?
38
Open Ended

Chromatography was performed on a sample of food colouring (‘X’) and 4 banned dyes (‘A’, ‘B’, ‘C’ and ‘D’). If X contains any of the 4 banned dyes, it is not safe to be consumed. What conclusions can we drawn from the chromatogram shown?
39

Identical dyes produce spots at the same height.
Sample X does not contain the banned dyes A, B and D.
However, X contains the banned dye C. Therefore, it must not be consumed.
Some text here about the topic of discussion.
Conclusions that can be drawn from the chromatogram:
40

Dyes A and D are pure.
Both dye B and dye C are mixtures of two different dyes.
Sample X is a mixture of three dyes.
Some text here about the topic of discussion.
Conclusions that can be drawn from the chromatogram:
41
Multiple Choice
If a substance is not soluble in the solvent, what happens to the spot placed on the start line?
a) Dot moves to the top of the paper
b) Dot does not move at all
c) Dot moves to the left or right
d) Dot moves down to the bottom of the paper
42
Multiple Choice
43
Multiple Choice
A pure substance shows ------------------ spot on chromatogram
0
1
2
3
44
Multiple Choice
45
Multiple Choice
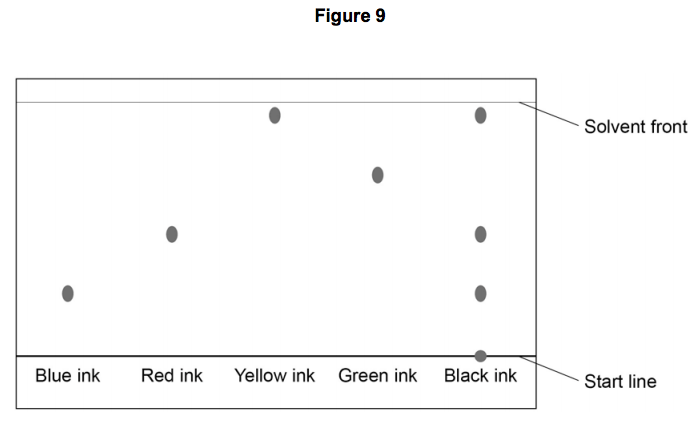
Referring to the chromatogram, black ink contains
Blue, red and yellow ink only
Blue, red, yellow and green ink
Blue and yellow ink
Black ink only
46
Multiple Choice

Which ink is a mixture?
Blue
Red
Yellow
Black
47
Multiple Choice

Which ink is most soluble?
Blue
Red
Yellow
Green
48
Multiple Select
What change of states are included in fractional distillation? (Can select two or more answers)
Melting
Evaporation
Condensation
Freezing
49
50
Multiple Choice
The laboratory apparatus used to collect 15.0 cm3 of oxygen gas from a chemical reaction.
gas syringe
condenser
pipette
burette
51
Multiple Choice
A liquid ester used to flavour food is believed to be impure. What would be the best way of testing its purity?
filter it
measure its boiling point
use paper chromatography
evaporation to dryness
52
Multiple Choice
In an experiment, a student needs to measure out 36.50 cm3 of a solution. Which piece of apparatus would measure this volume most accurately?
beaker
burette
measuring cylinder
pipette
53
Multiple Choice

An unknown gas can be collected using the apparatus shown.
Based on the diagram only, what can your infer about the physical property of this gas?
It is denser that air.
It is insoluble in water.
It is less dense than air.
It is soluble in water.
Separation Techniques

Show answer
Auto Play
Slide 1 / 53
SLIDE
Similar Resources on Wayground

49 questions
Unit 2 Cell Division R
Presentation
•
7th - 9th Grade

47 questions
Locomotor System
Presentation
•
7th - 9th Grade

46 questions
Ecology Part 1-4 Review
Presentation
•
6th - 9th Grade

46 questions
Lesson 2 Properties of Matter
Presentation
•
8th - 9th Grade

50 questions
C3.1 Chemical Formulae and Balancing Equations
Presentation
•
8th - 9th Grade

49 questions
Conflicts and Compromises (Pre-Civil War)
Presentation
•
6th - 9th Grade

49 questions
Contact and Noncontact Forces
Presentation
•
6th - 9th Grade

44 questions
Solar Eclipses vs. Lunar Eclipses
Presentation
•
6th - 10th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

34 questions
Unit 11 Acids and Bases Test Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

12 questions
IP PreAP: Fission and Fusion Reactions
Quiz
•
10th - 12th Grade

22 questions
Unit 8- Chemical Reactions
Quiz
•
10th Grade

34 questions
Nuclear Chemistry Review
Quiz
•
9th - 12th Grade