

Introduction to Solutions
Presentation
•
Science
•
8th - 12th Grade
•
Medium
Standards-aligned
B Taylor
Used 28+ times
FREE Resource
11 Slides • 31 Questions
1
Introduction to Solutions
Ms Taylor
2
Mixtures
A mixture contains two or more substances that are not chemically combined.
Can be:
Homogenous- A substance is homogeneous if its composition is identical wherever you sample it - it has uniform composition and properties throughout. Homogeneous is Latin for the same kind. ex, sea water
Heterogeneous - A mixture that does not have uniform composition and properties throughout.
3
Homogenous Mixture
Solution - a Homogenous mixture, usually of a solid and a liquid where the solid settles out, eg saltwater. The parts making up a solution keep their individual properties but the particles are not clearly seen or distinguished. It can be made from liquid in liquid;gas in gas; gas in liquid etc.
Consists of two parts:
Solute - What is dissolved in a solution eg, the salt in saltwater. Usually in smaller amount.
Solvent - Liquid in which something is dissolved, eg the water in saltwater. The larger part of the solution.
4
Multiple Choice
What is a substance that is dissolved in another substance?
solution
solute
solvent
compound
5
Multiple Choice
What is a solvent?
The substance that does the dissolving in a solution.
The substance that is being dissolved in a solution.
The mixing of different substances.
The process in which neutral molecules lose or gain electrons
6
Multiple Choice

Which substance is SOLUBLE in water?
sand
oil
salt (sodium chloride)
sulfur
7
Multiple Choice

Which substance is INSOLUBLE in water?
salt (sodium chloride)
sugar (sucrose)
oil
copper sulfate
8
Multiple Choice
salt water is a
compound
element
heterogeneous mixture
solution
9
Solubility
Solubility is how much solute dissolves in a certain amount of solvent at a certain temperature.
An Insoluble substance cannot be dissolved in the solvent.
Dissolve- "Like dissolves like'- polar solute dissolves in polar solvent and non-polar solute dissolve in non-polar solvent.(with reference to a solid) become or cause to become incorporated into a liquid so as to form a solution (solid particles are no longer visible).
Concentration - The amount of substance in a specified space. The more solute in the solvent, more concentrated. A small amount of solute in solvent makes a DILUTE solution.
10
Multiple Choice
When a Koolaid mix in water is dark in color and very sweet it is a __________ solution.
concentrated
Diluted
11
Multiple Choice
What does it mean to dilute a solution?
lower the concentration of solute per solvent
increase the concentration of solute per solvent
12
Factors that affect solubility
There are three factors that affect the solubility of a sulute. These are:
Temperature
Surface Area
Agitation/stirring
13
1. Temperature
The solubility of a given solute in a given solvent typically depends on temperature.
As the temperature of a solution is increased, the average kinetic energy of the molecules that make up the solution also increases.
This increase in kinetic energy allows the solvent molecules to more effectively break apart the solute molecules that are held together by intermolecular attractions.
Therefore, at higher temperatures, more of a solute is dissolved in the same amount of solvent. (ex. sugar dissolves easier in room temperature water than cold water)
14
Saturated and Unsaturated Solutions
An unsaturated solution is one that still has the ability to dissolve more solute particles.
A saturated solution is one that has dissolved the maximum amount of solutes possible at that temperature. A saturated solution will have few solute particles undissolved as that is the indication that it cannot dissolve any more solute
A super-saturated solution is one which has dissolved as much solute as possible at a higher temperature after it had already been saturated. These will form crystals when cooled.
15
2. Surface area
Surface Area - the outside part or uppermost layer of something (often used when describing its texture, form, or extent).
The SMALLER the particles, the GREATER the surface area that is exposed to the solvent.
Crushing the solute increases the amount of contact it has with the solvent. The particles of the crushed solute mix with the solvent more quickly and so dissolves faster.
Hence, clumps of sugar will take longer to dissolve than finer particles
16
3. Agitation/stirring
Agitation - the action of briskly stirring or disturbing something, especially a liquid.
Mixing by stirring or shaking causes the solute particles to separate from one another and spread out more quickly among the solvent particles as it helps to increase the surface area exposed to solvent particles.
17
Multiple Choice

How does a solution become supersaturated?
dissolve lots of solute in it.
dissolve a little solute in it.
dissolve more solute than you should be able to.
dissolve a solvent in it.
18
Multiple Choice
When a certain amount of solvent cannot hold any more solute it is called a ________ solution.
Diluted
Saturated
19
Multiple Choice
When materials combine to form a mixture, they
A. keep their original properties.
B. react to form a new substance with new properties.
C. combine in a specific ratio.
D. always change their physical state.
20
Multiple Choice
When a solvent contains as much of the solute as it can hold, the solution is said to be
supersaturated
diluted
saturated
unsaturated
21
Multiple Choice
Stirring the solution increases or decreases the solubility?
increases
decreases
22
Multiple Choice
Solution where more solute can still be dissolved at the given temperature.
Saturated
Unsaturated
Supersaturated
Homogeneous solution
23
Multiple Choice

Which factor is shown in the picture?
temperature
particle size
stirring
use of spoon
24
Multiple Choice
Why do sugar particles dissolve faster in hot water?
water particles move slow
water particles move fast
water particles settle down
water particles stay on top
25
Multiple Choice
What combination would dissolve a solid solute the fastest?
high temperature, no stirring
no heat, no stirring
sugar cube, no heat
high temperature, stirring
26
Multiple Choice

The beaker on the left is _______________ and the beaker on the right is _______________
Concentrated, dilute
Dilute, concentrated
Soluble, insoluble
Insoluble, soluble
27
Multiple Choice
Which of the following does not affect solubility of solutes?
Tempearture
Color
Size particles
Kind of solute
28
Multiple Choice
Which of the following statements is true about how using smaller salt crystals would affect the rate of making a salt solution in water?
Smaller crystals increase the surface area and slow down dissolving.
Smaller crystals decrease the surface area and speed up dissolving.
Smaller crystals increase the surface area and speed up dissolving.
Smaller crystals decrease the surface area and slow down dissolving.
29
Multiple Choice
Which will dissolve faster crushed table salt or crystal of table salt?
crushed table salt
crystal of table salt
30
Multiple Choice
The concentration of a mixture can be increased in which of the following ways?
Heating the mixture
Adding more water “solvent”
Adding more powder “solute”
Stirring the mixture
31
Multiple Choice
Which of the following actions will NOT
increase the rate of dissolution
(dissolving) for solids?
Stirring the solution
Decreasing the temperature
Increasing the surface area of the
solute
Increasing the temperature
32
Multiple Choice
Which of the following actions will NOT
increase the rate of dissolution
(dissolving) for solids?
Stirring the solution
Decreasing the temperature
Increasing the surface area of the
solute
Increasing the temperature
33

These are used to map the state of solubility of a substance AT A GIVEN TEMPERATURE.
IF the amount of solute in the solution at a STATED temperature falls:
on the line- it is saturated at that temperature
Under the line-unsaturated at that temperature
Above the line-supersaturated at that temperature
Solubility Charts
34

Usually the graphs shows solubility Curves for more than one substance.
Select the curve that matches the substance you need and ignore the others.
Note points to the left of graph!!
Reading Solubility Graphs
35
Multiple Choice

36
Multiple Choice

37
Multiple Choice

38
Multiple Choice

How many grams of SO2 can dissolve at 50 ⁰C in 100 g of water?
5 g
10 g
20 g
39 g
39
Multiple Choice
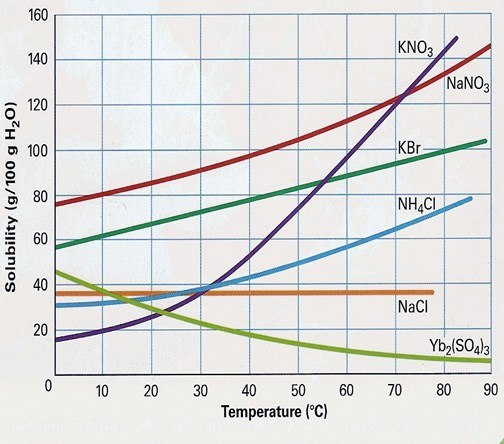
At 80'C, KBr's solubility is:
100
90
80
0
40
Multiple Choice

How much NaBr solute can be dissolved at 70 degrees?
110
120
130
140
41
Multiple Choice

When 40 grams of NaCl is dissolved in 100 grams of water at 90 ºC, the solution can be correctly described as:
supersaturated
saturated
unsaturated
42
Multiple Choice

What is the solubility of NaNO3 at 20 C in 100 g of water?
90 g
80 g
40 g
160 g
Introduction to Solutions
Ms Taylor
Show answer
Auto Play
Slide 1 / 42
SLIDE
Similar Resources on Wayground

38 questions
Chemical Reactions - Types of Reactions
Presentation
•
9th - 12th Grade

35 questions
Regulating the Cell Cycle + Cancer (part 3)
Presentation
•
9th - 12th Grade

35 questions
Nuclear fission vs fusion
Presentation
•
9th - 12th Grade

34 questions
Read About It: Ecosystem Disruptions
Presentation
•
8th Grade - University

40 questions
Colonial Regions 2025-2026
Presentation
•
8th - 12th Grade

38 questions
Unit 4- Lesson 4: Elections
Presentation
•
9th - 12th Grade

38 questions
Proving Triangles are Congruent
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

40 questions
NCFE Earth and Environmental Science Released Test
Quiz
•
9th - 12th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

19 questions
Renewable and Nonrenewable Resources (CK)
Quiz
•
8th Grade

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade