

RATE OF REACTION
Presentation
•
Chemistry
•
11th Grade
•
Medium
SITI Moe
Used 7+ times
FREE Resource
18 Slides • 34 Questions
1
RATE OF REACTION
by SITI Moe

2



3
Example of reaction
4
Example of reaction
5
Example of reaction
6
Example of reaction
7
Example of reaction
8
Multiple Choice
Choose the correct answer for the fast reaction
fermentation of glucose solution
photosynthesis
iron rusting
combustion
9
Multiple Choice
What is the best answer for slow reaction
reaction between acid and alkali
Iron rusting
combustion
double decomposition
10
Multiple Choice
Which of the following reactions occurs at the highest rate?
Photosynthesis
Rusting of iron
Combustion of magnesium in oxygen
11
Open Ended
What do you understand by the rate of reaction?
12

13
Multiple Select
Which of the following is/are the unit(s) for rate of reaction?
cm3s−1
cm2s−1
cm s−1
kg min−1
mol s−1
14
Multiple Choice
Which of the following has the lowest rate of reaction?
Combustion of ethanol
Fermentation of glucose
Oxidation of magnesium
Precipitation of silver chloride
15
Multiple Choice
Which reactions has the highest rate of reaction?
Rusting of water pipe
Photosynthesis in green plant
Burning of a small piece of charcoal in the air
Formation of stalactites and stalagmites
16
Multiple Choice
Which process has the highest rate of reaction?
Rusting
Respiration
Combustion
Photosynthesis
17
Multiple Choice
What is the meaning of the rate of reaction?
Decrease in amount of product
Decrease in amount of product against time
Increase in amount of products against time
Increase in amount of reactants against time
18
Multiple Choice
Which unit is correct for the rate of reaction?
g mol-1
g min-1
mol dm-3
kJ mol-1
19
Multiple Choice
The following equation shows the reaction between calcium carbonate, CaCO3 and hydrochloric acid, HCl:
CaCO3(aq) + 2HCl(aq) → CaCl2(aq) + CO2 (g) + H2O(l)
Which of the following is the suitable method to determine the rate of reaction?
Change in the temperature of the solution with time
Change in the volume of carbon dioxide gas with time
Change in the mass of water with time
Change in the concentration of hydrochloric acid with time
20
Determining rate of reaction must be made based on the changes that are observable and can be measured in a certain period of time. What are these changes?

Formation of precipitate
21


Decrease in mass of reactant
Increase in volume of gases
22
Multiple Choice
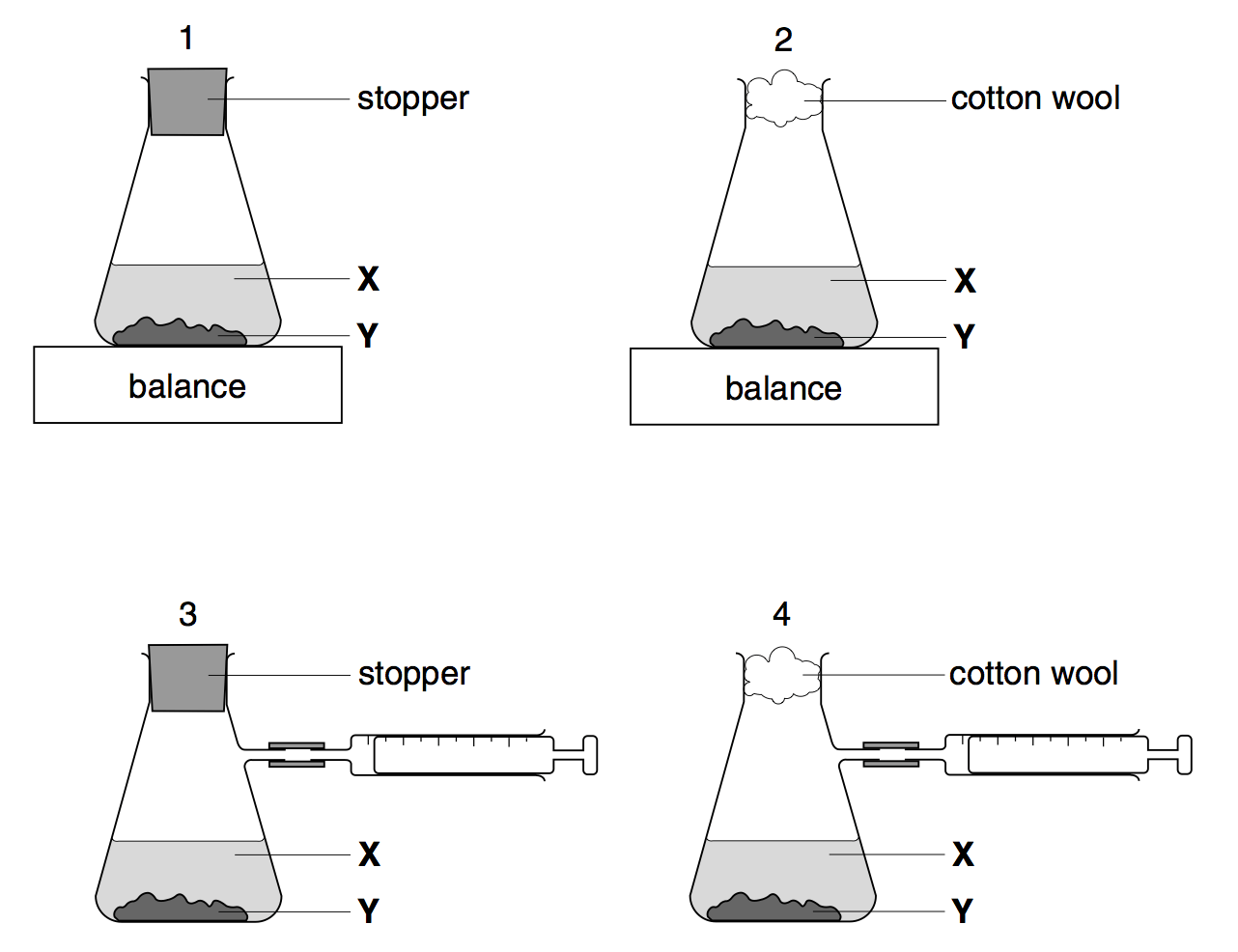
Which two diagrams show suitable methods for investigating the speed of the reaction?
23
Multiple Select
This method can use to collect gas produced ...
using a gas syringe system
downwards water displacement
using digital balance
24
Multiple Choice
The following equation represents the reaction between calcium carbonate, CaCO3 and hydrochloric acid, HCl.
CaCO3 + 2HCl --> CaCl2 + CO2 + H2O
Which changes can be used to determine the rate of reaction?
I : mass of calcium carbonate per unit time
II : Volume of carbon dioxide released per unit time
III : Colour of solution per unit time
IV : Mass of precipitate produced per unit time
I and II
I and III
II and IV
III and IV
25
Multiple Choice
The following equation shows the reaction between calcium carbonate, CaCO3 and hydrochloric acid, HCl:
CaCO3(aq) + 2HCl(aq) → CaCl2(aq) + CO2 (g) + H2O(l)
Which of the following is the suitable method to determine the rate of reaction?
Change in the temperature of the solution with time
Change in the volume of carbon dioxide gas with time
Change in the mass of water with time
Change in the concentration of hydrochloric acid with time
26
Multiple Choice
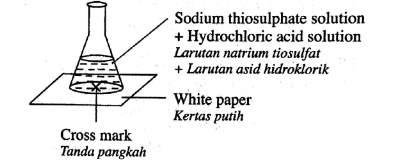
Diagram shows the apparatus set up for an experiment to determine the rate of reaction.
Which of the following techniques is the most suitable to determine the rate of reaction?
Record the time as soon as precipitate is formed
Record the time taken to obtain the maximum temperature
Record the times as soon as the cross mark cannot be seen
Record the times taken for the change of the pH value until a fixed pH value is obtained
27
Multiple Choice
The reaction between zinc, Zn and hydrochloric acid, HCl is represented by the following equation.
Zn + 2HCl --> ZnCl2 + H2
A student wants to determine the rate of reaction in a school laboratory. Which of the following methods is the most suitable?
Determine the change in temperature of the solution with time
Determine the change in the concentration of zinc chloride with time
Determine the volume of hydrogen gas given off with time
Determine the change in the concentration of hydrochloric acid with time
28
Determining the time for reaction with reference to some observable and measurable changes.

A) Reaction between zinc, Zn and sulphuric acid, H2SO4
29
Multiple Select

State the observable change?
Change in electrical conductivity
Increase in the volume of gases
Change in pH Value
Change in pressure
30
Open Ended

How would you know the reaction completed?
31
Determining the time for reaction with reference to some observable and measurable changes.
B) Reaction between nitric acid, HNO3 and marble chips, CaCO3

32
Multiple Select

State the observable change?
Change in electrical conductivity
Decrease of the mass of the reactant
Increase in the volume of gases
Change in pH Value
Change in pressure
33
Multiple Select

State is the products obtain from this reaction?
Salt
water
hydrogen gas
carbon dioxide gas
34
Open Ended

How do you know that the reaction completed?
35
Determining the time for reaction with reference to some observable and measurable changes.
c) Reaction between potassium iodide solution, KI and lead(II) nitrate solution, Pb(NO3)2

36
Multiple Select

State the observable change?
Change in electrical conductivity
Decrease of the mass of the reactant
Increase in the volume of gases
Formation of precipitate
Change in pressure
37
Multiple Select

What is the colour of precipitate form
Brown
White
Yellow
38
Average Rate of Reaction
Instantaneous Rate of Reaction
Type of Rate Of Reaction
39

40

41

42
Multiple Choice

Table 4 shows the total volume of gas evolved at different intervals for the dissociation of hydrogen peroxide.
What is the average rate of reaction in the second minute?
14.0 cm3 min-1
16.0 cm3 min-1
18.0 cm3 min-1
28.0 cm3 min-1
43
Multiple Choice

Table 4 shows the total volume of gas evolved at different intervals for the dissociation of hydrogen peroxide.
What is the average rate of reaction in the second minute?
14.0 cm3 min-1
16.0 cm3 min-1
18.0 cm3 min-1
28.0 cm3 min-1
44
Multiple Choice

Table 7 shows the volume of carbon dioxide gas collected in an experiment for the reaction between excess calcium carbonate and hydrochloric acid.
What is the average rate of reaction for the experiment?
0.12 cm3 s-1
0.18 cm3 s-1
0.22 cm3 s-1
0.37 cm3 s-1
45
Multiple Choice

Table 19 shows the volume of gas collect for each minute for one reaction.
what is the rate of reaction for the first 4 minute of this reaction?
0.017 cm3 s-1
0.111 cm3 s-1
0.133 cm3 s-1
0.150 cm3 s-1
46
Multiple Choice
In a reaction between magnesium carbonate and sulphuric acid, the following volume of carbon dioxide is collected for the 10 minutes period of reaction, with each reading taken every minute interval. 0 minute: 0 cm3 1 minute: 25 cm3 2 minute: 40 cm3 3 minute: 51 cm3 4 minute: 58 cm3 5 minute: 63 cm3 6 minute: 63 cm3 7 minute: 63 cm3 Calculate the rate of reaction from the 2 minute to 5 minute.
12.50 cm3min−1
7.67 cm3 min−1
12.60 cm3min−1
20.00 cm3min−1
47

The instantaneous rate of reaction is the rate of reaction at a particular point of time. It is determined from the experimental data by plotting a graph

48
Multiple Choice
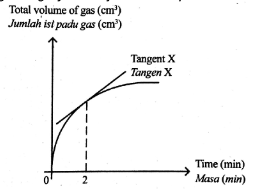
Diagram shows a graph to study the effect of total surface area on the rate of reaction.
What is represented by gradient tangent X?
Average rate of reaction in the first 2 minutes
Average rate of reaction in the 2nd minute
Overall average rate of reaction
Rate of reaction at the 2nd minute
49
Multiple Choice

Diagram 4 shows the graph of volume of carbon dioxide gas against time when 5 g of marble chips is added to
50 cm3 of 0.2 mol dm-3 hydrochloric acid.
At what time the rate of reaction the highest?
t1
t2
t3
t4
50
Multiple Choice

Which of the curves indicates the slowest reaction?
A
B
C
51
Multiple Choice

Which of the curves indicates the fastest reaction?
A
B
C
52
Multiple Choice
The equation represents the reaction between sodium carbonate and hydrochloric acid.
Na2CO3 + 2 HCl → 2 NaCl + H2O + CO2
The mass of the beaker and its contents is plotted against time.
Which graph represents what happens when sodium carbonate reacts with an excess of dilute hydrochloric acid?
RATE OF REACTION
by SITI Moe

Show answer
Auto Play
Slide 1 / 52
SLIDE
Similar Resources on Wayground

46 questions
SCH3U: Unit #2 Review
Presentation
•
11th Grade

45 questions
Unit 3 Kinetic Molecular Theory
Presentation
•
11th Grade

45 questions
Unit 5 - Reactions
Presentation
•
11th Grade

47 questions
How a Bill Becomes a Law
Presentation
•
11th - 12th Grade

51 questions
Unit 2: Lesson 7 Half Life Decay
Presentation
•
10th - 12th Grade

45 questions
Measurements in Science and Sci Notation
Presentation
•
10th - 12th Grade

44 questions
Unit 2 Lesson 4 Atomic Theory and PEN
Presentation
•
10th - 12th Grade

47 questions
Development of Atomic Theory
Presentation
•
11th Grade
Popular Resources on Wayground

25 questions
The Ultimate College Knowledge Quiz
Quiz
•
8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Math Review
Quiz
•
6th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade