

from wk15 quiz atoms, neutrons, protons, isotopes
Presentation
•
Science
•
9th Grade
•
Medium
. Ade
Used 4+ times
FREE Resource
4 Slides • 38 Questions
1
wk15 Thur 11/11/21
by . Ade
2
Multiple Choice

3
Multiple Choice

4
Multiple Choice

5
Multiple Choice

6
Multiple Choice
Mass minus Atomic Number Equals (M-A=?)
Neutrons
Protons
Nucleus
Atom
7
Multiple Choice

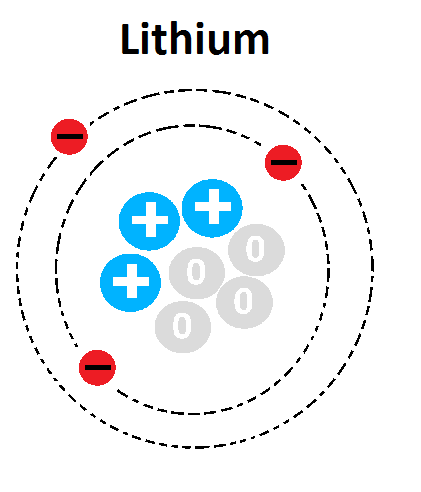
What goes in place of the question marks?
Neutrons
Electron Cloud
Atomic Number
None of the above
8
Multiple Choice

9
Multiple Choice
10
Multiple Choice
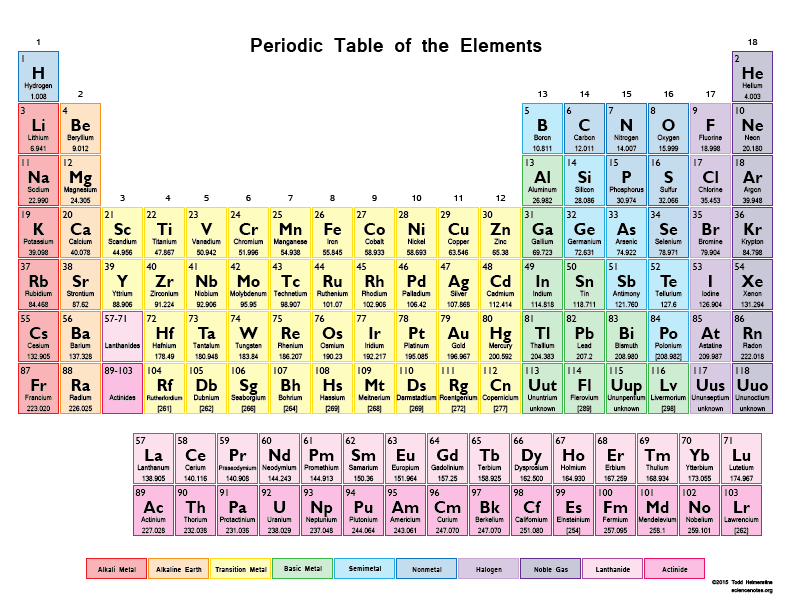
How many protons are in a sodium atom, Na? (tap to enlarge the periodic table)
Sodium has 1 proton.
Sodium has 3 protons.
Sodium has 11 protons
11
Multiple Choice

12
Multiple Choice
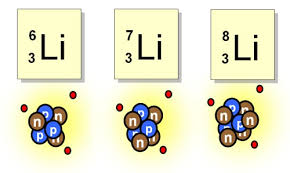
13
Multiple Choice
14
Multiple Choice
15
Multiple Choice
16
Multiple Choice

17
Multiple Choice
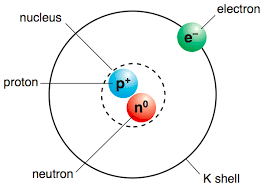
18
Multiple Choice

19
Multiple Choice

20
Multiple Choice
21
Multiple Choice
22
Multiple Choice

23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice

27
Multiple Choice

28
How to:
Calculate an atom's mass
Calculate the neutrons in an atom

By: Ms. LaLonde-Bernal
29
To calculate the mass of an atom you add the protons and the neutrons.
Atomic mass = protons + neutrons

30
Multiple Choice

31
Fill in the Blanks
Type answer...
32
Fill in the Blanks
Type answer...
33
Fill in the Blanks
Type answer...
34
Fill in the Blanks
Type answer...
35
To calculate the number neutrons in an atom. Subtract the number of protons (the atomic number) from the atomic mass (the number of protons + neutrons).
Neutrons = Atomic mass - protons

36
Multiple Choice

How many neutrons are in one atom of the element shown?
ROUND the atomic mass to the nearest whole number.
Example: 26.982 rounds to 27
27
13
26.98
14
37
Multiple Choice

How many neutrons are in one atom of the element shown?
ROUND the atomic mass to the nearest whole number.
Example: 26.982 rounds to 27
30
26
55.847
56
38
Multiple Choice

How many neutrons are in one atom of the element shown?
ROUND the atomic mass to the nearest whole number.
Example: 26.982 rounds to 27
28
57
58.69
31
39
Multiple Choice

How many neutrons are in one atom of the element shown?
ROUND the atomic mass to the nearest whole number.
Example: 26.982 rounds to 27
3
7
4
6.94
40
Multiple Choice

How many neutrons are in one atom of the element shown?
ROUND the atomic mass to the nearest whole number.
Example: 26.982 rounds to 27
26
30
55
55.85
41
Multiple Choice

42
Multiple Choice
wk15 Thur 11/11/21
by . Ade
Show answer
Auto Play
Slide 1 / 42
SLIDE
Similar Resources on Wayground

41 questions
meiosis
Presentation
•
9th Grade

33 questions
Electromagnetic Waves
Presentation
•
9th Grade

38 questions
Properties of Matter
Presentation
•
8th Grade

36 questions
Bohr Diagrams
Presentation
•
8th Grade

39 questions
INTERNAL STRUCTURE OF THE EARTH
Presentation
•
8th - 10th Grade

35 questions
Stars / HR Diagrams
Presentation
•
8th Grade

37 questions
Photosynthesis/Cellular Respiration
Presentation
•
9th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

40 questions
NCFE Earth and Environmental Science Released Test
Quiz
•
9th - 12th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

61 questions
End of the Year Trivia Fun
Quiz
•
9th Grade

15 questions
Protein synthesis
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade