

APT Test Review
Presentation
•
Science, Physics, Chemistry
•
6th - 8th Grade
•
Medium
Standards-aligned

Jawara Senghor
Used 36+ times
FREE Resource
15 Slides • 31 Questions
1
by Mr. Senghor

2
The Atom
An atom is the smallest particle into which an element can be divided and still be the same substance. All atoms of a particular element are identical but are different from atoms of all other elements. Every atom of silver, for example, is the same as every other atom of silver but different from an atom of iron.

3
Parts of an atom
The Nucleus
Protons are the positively charged particles within the atom. They have a mass of of 1u. Neutrons are particles that carry no electric charge and have the same mass as a proton (1u). Together, protons and neutrons form the nucleus of the atom located in its center.

4
Parts of an atom
The electron cloud
Electrons are the negatively charged particles in the atom that move around the nucleus at almost the speed of light. Scientists have found that it is impossible to determine the exact position of an electron and for that reason, the electrons are pictured as being in a cloud around the nucleus.

5

6
Multiple Choice
What are the 3 subatomic particles of an atom?
carbon atom, proton, electron
quarks, neutrons, protons
nucleus, protons, electrons
protons, neutrons, electrons
7
Multiple Choice
Which subatomic particle has a negative charge?
proton
neutron
electron
8
Multiple Choice
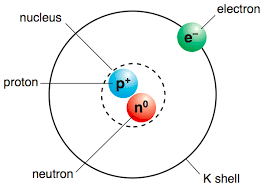
Which subatomic particles contribute the most to the mass of an atom?
Protons and Neutrons
Protons only
Electrons only
9
Multiple Choice

10
Multiple Choice

Which subatomic particles contribute the least to the mass of an atom?
Neutrons
Protons
Electrons
11
Identifying Atoms
By atomic number - The number of protons in the nucleus of an atom is the atomic number of that atom. The number of protons distinguishes the atoms of one element from the atoms of another. For example, every hydrogen atom contains one proton while every carbon atom has exactly six protons in its nucleus.
By mass number - The combined number of protons and neutrons in an atom's nucleus is its mass number. While the atoms of a certain element always has the same number of protons, they may not always have the same number of neutrons. Atoms of the same element that have a different number of neutrons are called Isotopes.
12

13
Multiple Choice
14
Multiple Choice
15
Multiple Choice
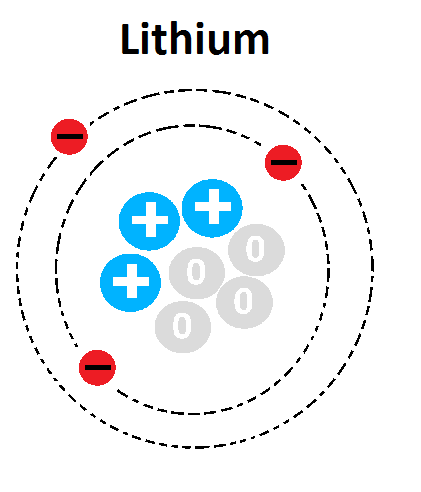
16
Multiple Choice
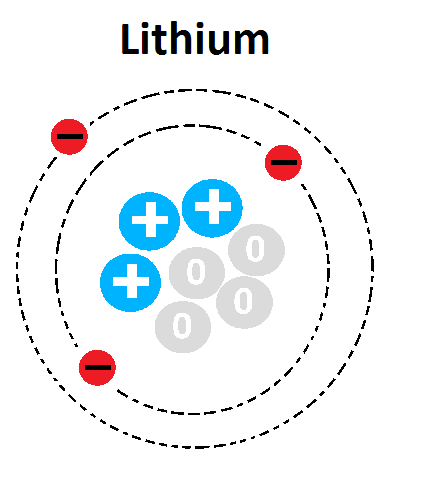
What is the mass number of this atom?
1
3
4
7
17
The Periodic Table
The periodic table of the elements, in chemistry, is the organized array of all the chemical elements in order of increasing atomic number. When the chemical elements are thus arranged, there is a recurring pattern called the “periodic law” in their properties, in which elements in the same group have similar properties. The initial discovery, made by Dmitri I. Mendeleev in the mid-19th century, has been of immense value in the development of chemistry.

18
Square Biz
The Periodic Table is not simply a list of the names of a bunch of elements, it contains useful information about each of them. The table is shown as a grid of squares, with each square containing the element's atomic number, chemical symbol, element name and average atomic mass.

19

20
Multiple Choice

Where is the element's atomic NUMBER located?
the top
the bottom
the top + the bottom
the number of protons + the number of neutrons
21
Multiple Choice

22
Multiple Choice

23
Regions of the Periodic Table
Elements of the periodic table can be classified into three major categories: metals, nonmetals and metalloids. There is a zig-zag line that looks like a staircase can help you identify where these elements are located. The elements bordering the zig-zag line are metalloids. All of the elements to the left of the zig-zag line are metals, except for hydrogen. Nonmetals are to the right of the metalloids.

24
Groups & Periods
Every vertical column of elements is called a group. In total, there are 18 groups on the periodic table. Elements in the same group often have similar physical and chemical properties and for this reason, a group is sometimes called a family. The elements in a group are similar because they have the same number of valence electrons.
Each horizontal row of elements on the periodic table is called a period. The physical and chemical properties of elements change in predictable ways from one end of the period to another. Atomic size increases as you move from left to right. Also, elements in the same period have the same number of electrons shells.
25

26
Multiple Choice
how many groups/families are on the table
15
16
17
18
27
Multiple Choice
what is the biggest group on the table
metals
nonmetals
metaloids
halogens
28
Multiple Choice
how many rows are on the table
4
5
6
7
29
Multiple Choice
where are the metaloids found
far left, vertical
stair step/diagonal
far right horizontal
at the bottom
30
APE MAN
In order to figure out the structure of an atom, you can use the acronym APE MAN along with the periodic table. The acronym is as follows
Atomic Number =
Protons =
Electrons
Mass number -
Atomic number =
Neutrons
31

32
Multiple Choice
33
Multiple Choice
34
Multiple Choice

35
Multiple Choice
What is the number of protons in the nucleus of an atom that determines the chemical properties of an atom?
mass number
atomic mass
atomic number
atomic symbol
36
Multiple Choice
What is the combined mass of all the protons and neutrons of an atom and is approximately equal to the number of protons and neutrons?
atomic mass
atomic number
subatomic particles
nucleus weight
37
Multiple Choice
how many rows are on the table
4
5
6
7
38
Multiple Choice
what is the small group of elements called that include C,N,O
metals
nonmetals
metaloids
noble gases
39
Multiple Choice
40
Multiple Choice
41
Multiple Choice

42
Multiple Choice
43
Multiple Choice
44
Multiple Choice

45
Multiple Choice

46
Multiple Choice
What do the horizontal rows on the periodic table tell us?
The number of orbitals
The number of valence electrons (the electrons on the outer most shell)
by Mr. Senghor

Show answer
Auto Play
Slide 1 / 46
SLIDE
Similar Resources on Wayground

40 questions
Hydrosphere Unit Review
Presentation
•
6th - 8th Grade

39 questions
Test Term 2 Review (2021)
Presentation
•
6th - 8th Grade

38 questions
Science 7 Year End Review - Unit B
Presentation
•
7th Grade

41 questions
Atoms and Elements
Presentation
•
6th - 8th Grade

45 questions
Life Cycle of a Star
Presentation
•
KG - 8th Grade

42 questions
CH 0 Understanding Science
Presentation
•
6th - 8th Grade

42 questions
RFMS Science: KE Calculations Review Lesson
Presentation
•
6th - 8th Grade

38 questions
Topographic Maps
Presentation
•
6th - 8th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

45 questions
Food Webs
Quiz
•
6th - 8th Grade

50 questions
Stars, Galaxies, HR Diagram
Quiz
•
8th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

20 questions
Abiotic & Biotic Factors
Quiz
•
7th Grade

40 questions
Unit F1 Review
Quiz
•
8th Grade