

Thermodynamics
Presentation
•
Physics
•
9th - 12th Grade
•
Medium
+4
Standards-aligned
Stacy King
Used 163+ times
FREE Resource
17 Slides • 21 Questions
1
Thermodynamics
By Stacy King
2

A measurement that describes the average kinetic energy in random thermal motion per atom or molecule
Temperature
3
Multiple Choice
4

A calibration tool to measure temperature changes
3 different scales
Fahrenheit
Celsius
Kelvin- the base unit of temperature in the International System of Units, having the unit symbol K
Thermometers
5
Multiple Choice
6
is produced when a rise in temperature causes atoms and molecules to move faster and collide with each other
Thermal Energy

7
Multiple Choice

8

the energy associated with the random, disordered motion of molecules within the system
ENTROPY
the degree of disorder or randomness in the system
Internal Energy
9
Multiple Choice
10

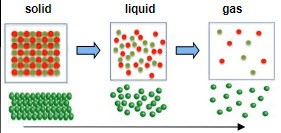
The more disorganized the molecules are the higher state of entropy
Entropy increases as temperature increases
Entropy and States of Matter
11
Multiple Choice
Do molecules generally move faster at 45 C or 106 C?
45 C because molecules have more energy than in 106C
106 C because molecules have more energy than in 45C
106 C because molecules have less energy than in 45C
45C because molecules have less energy than in 106 C
12
Multiple Choice
13

All kinetic energy stops
the minimum possible
temperature, 0 K, or −273ºC (−459ºF)
Absolute Zero
14
Multiple Choice
What happens at absolute zero?
All Motions cease
everything turns to a solid
everything dies
who knows?
15

Changes both the temperature and the state of matter
Travels from high to low
Heat
16
0th Law
Thermal equilibrium
The condition under which two substances in physical contact with each other exchange no heat energy
temperatures balance out, heat stops flowing, then the system (or set of systems)
Laws of Thermodynamics

17
Multiple Choice
18
Multiple Choice

19
1st Law
Conservation of Energy
Energy cannot be created or destroyed only changed into another form
Laws of Thermodynamics

20
Multiple Choice
21
2nd Law
Heat
Heat always flows from hot to cold
Laws of Thermodynamics

22
Multiple Choice
23
Multiple Choice
24
3rd Law
Entropy
As temperature approaches absolute zero, entropy is at constant minimum
Laws of Thermodynamics

25
Multiple Choice
26
Multiple Choice
27

Conduction
the transfer of heat through or between materials by direct contact
Heat Transfer Method
28
Multiple Choice
Conduction
The transfer of thermal energy through solids
The transfer of thermal energy through liquids and gases
The transfer of thermal energy as electromagnetic waves
29

Convection
the transfer of heat by the motion of liquids and gases
Heat Transfer Method
30
Multiple Choice
Convection
The transfer of thermal energy through liquids and gases
The transfer of thermal energy through solids
The transfer of thermal energy as electromagnetic waves
31
Multiple Choice
32

Radiation
the dispersal of energy through space through electromagnetic waves
Heat Transfer Method
33
Multiple Choice
34
Multiple Choice
Radiation
The transfer of thermal energy as electromagnetic waves
The transfer of thermal energy through solids
The transfer of thermal energy through liquids and gases
35
materials that transfer heat readily (ex: diamond, copper, aluminum, gold)
Thermal Conductors

36
materials that do not transfer heat readily (ex: plastic, wool, styrofoam)
Thermal Insulators

37
Multiple Choice
38
Multiple Choice
Heat moves easily through materials called ____
Wood
Plastic
Rubber
Metal
Thermodynamics
By Stacy King
Show answer
Auto Play
Slide 1 / 38
SLIDE
Similar Resources on Wayground

35 questions
Telling time in Spanish
Presentation
•
9th - 12th Grade

35 questions
Charles Darwin and Evolution Lesson
Presentation
•
9th - 12th Grade

35 questions
Reflection and refraction
Presentation
•
9th - 11th Grade

33 questions
Incline Forces: Review
Presentation
•
9th - 12th Grade

34 questions
Solving Radical Equations
Presentation
•
9th - 12th Grade

34 questions
Macromolecules
Presentation
•
9th - 12th Grade

35 questions
Introduction to Bonding
Presentation
•
9th - 12th Grade

35 questions
Saber v Conocer
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Physics

11 questions
Electricity Explained
Interactive video
•
9th - 12th Grade

18 questions
Series and Parallel Circuits
Quiz
•
9th Grade

15 questions
waves and wave properties
Quiz
•
9th - 12th Grade

35 questions
Electricity and Magnetism SPS10
Quiz
•
8th - 10th Grade

10 questions
Exit Check 7.1 Intro to Waves
Quiz
•
9th Grade

10 questions
Exit Check 7.2: Electromagnetic Waves
Quiz
•
9th Grade

10 questions
Exit Check 7.3 - Analog and Digital
Quiz
•
9th Grade

18 questions
Unit 1 and 2 Concepts Review
Quiz
•
9th - 12th Grade