

G5-Term2 Final Review Part1
Presentation
•
Science
•
5th Grade
•
Medium
Mai Hamza
Used 5+ times
FREE Resource
5 Slides • 33 Questions
1
G5-Term2 Final Review Part1
By Mai Hamza
2

distinguish the parts of atoms
Investigate the properties of metals, nonmetals and metalloids
Atom: smallest unit
I can
3
Poll
Are You Ready?
4

smallest unit of the element
Inside nucleus: Protons and Neutrons
Outside nucleus: Electron
What Should I know about Atoms?
5
Draw
Complete the labels
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice
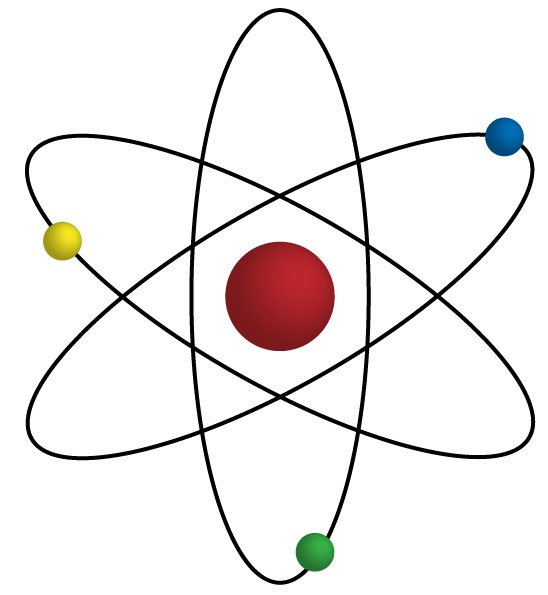
11
Multiple Choice
The smallest particles of matter are called
neutrons
atoms
electrons
protons
12
Multiple Choice
13

118 elements
arranged in periodic table
Metals - metalloids and nonmetals
What Should I know about elements?
14
Multiple Choice
How many types of elements are there?
1
2
3
4
15
Multiple Choice
Which types of elements that their texture can be shiny.
Non-metals
Non-metals and Metalloid
Metal and Non-metal
Metal and Metalloid
16
Multiple Choice
Carbon, phosphorus, sulfur, and bromine are .....
metals
non-metals
metalloids
17
Multiple Choice
Boron, silicon, germanium, arsenic, and antimony are ....
metals
non-metals
metalloids
18
Multiple Choice

19
Multiple Choice

20
Multiple Choice

21
Multiple Choice

22
Multiple Choice

23
Multiple Choice

24
Multiple Choice

How many different elements are there?
10
50
100
118
25
Element Key
Atomic Number
Atomic Mass
Protons
Neutrons
Electrons
Name
What Should I know about elements?

26
Multiple Choice

Symbol
Atomic Weight
Common Name
Atomic Number
27
Multiple Choice
What Determines an Element's Atomic Number?
Protons + Neutrons
Electrons - Protons
Number of Protons in the Nucleus
Number of Neutrons in the Nucleus
28
Multiple Choice

Atomic Number
Atomic Symbol
Common Name
Atomic Weight
29
Multiple Choice

Atomic Number
Atomic Weight
Atomic Symbol
30
Multiple Choice
How Do Scientists Determine the Atomic Weight?
Protons + Neutrons
Number of Electrons
Neutrons + Electrons
Number of Protons in the Nucleus
31
Multiple Choice

What is this Element's Atomic Number?
20
10
6
30
32
Multiple Choice

What Is This Element's Common Name?
Neon
Ne
English
Latin
33
Multiple Choice

How Many Neutrons Does This Element Have?
30
10
20
5
34
Multiple Choice

What Is This Element's Symbol
Ne
Neon
N
35
Multiple Choice

How many protons does this element have?
20
10
3
5
36
Multiple Choice

What is this Element's atomic weight/mass?
20.1797
30
10
5
37
Multiple Choice

What is this element's common name?
Neon
Ne
Neat
No
38
Poll
Did you like today's session?
G5-Term2 Final Review Part1
By Mai Hamza
Show answer
Auto Play
Slide 1 / 38
SLIDE
Similar Resources on Wayground

29 questions
Bozman 6th Grade Science CA 1 Review
Presentation
•
6th Grade

36 questions
Forms of Energy
Presentation
•
5th Grade

32 questions
Journey to the Center of the Earth
Presentation
•
6th Grade

34 questions
Energy
Presentation
•
6th Grade

28 questions
Properties of Matter Practice Test
Presentation
•
5th Grade

29 questions
The Way We See Things
Presentation
•
5th Grade

39 questions
Matter
Presentation
•
5th Grade

31 questions
5.1.11 Live
Presentation
•
5th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

20 questions
Science Review
Quiz
•
5th Grade

20 questions
Food Chains and Food Webs Review
Quiz
•
5th Grade

45 questions
Science SOL Review 1
Quiz
•
5th Grade

118 questions
5th Grade SSA Review_Mr. Hite
Quiz
•
3rd - 5th Grade

50 questions
5th Grade Science EOG Review
Quiz
•
5th Grade

43 questions
4th and 5th Grade VA Science SOL Review
Quiz
•
5th Grade

34 questions
5th Grade MAP Science Session 2 Released
Quiz
•
5th Grade

20 questions
Life Science Review
Quiz
•
5th Grade