

HEAT and TEMPERATURE
Presentation
•
Science
•
9th Grade
•
Medium
+2
Standards-aligned
Roberto Gonzalez
Used 16+ times
FREE Resource
8 Slides • 21 Questions
1
HEAT and TEMPERATURE
By Roberto Gonzalez
2
3
Open Ended
How would you describe or define the term "Heat"?
4
5
Multiple Choice
Kinetic energy refers to......
Work
Strength
Warming
Movement
6
7
Multiple Choice
Energy of motion
Thermal
Stored
Kinetic
Potential
8
Multiple Choice
When you heat up an object, the particles in the object move _________.
faster
the same speed
slower
9
10
Multiple Choice
11
Fill in the Blanks
Type answer...
12
Multiple Choice
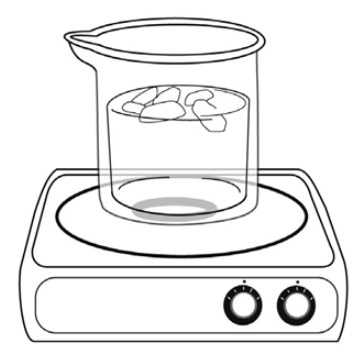
Emerson places a beaker of water on a hotplate and turns the hotplate on. The temperature in the room is 25 ºC. After the water heats to 75 ºC, she adds pieces of ice to the beaker.
Which of the following describes a transfer of heat energy that occurs during this experiment?
from the air to the water
from the ice to the beaker
from the water to the ice
from the water to the hotplate
13
Multiple Choice

Jeriah places two metal cubes in contact with each other. Energy transfers as heat flows from Cube A to Cube B. What conclusion can you draw regarding Cube A?
It is larger than Cube B.
It is cooler than Cube B.
It is smaller than Cube B.
It is warmer than Cube B.
14
Multiple Choice
How does heat transfer when you hold an ice cube in your hand?
from the cold ice cube to your warm hand
the cold ice cube makes your hand cold
from your warm hand to the cold ice cube
very carefully
15
Multiple Choice
16
Multiple Choice
If two objects have different temperatures, heat will flow from the warmer object to the cooler one UNTIL ____________
one reaches a temperature of zero
they both have an equal temperature
one runs out of energy
17
18
Multiple Choice

19
Multiple Choice

20
Multiple Choice
21
Multiple Choice
Measure of the average kinetic energy of a substance
Total Thermal Energy
Heat Exchange
Temperature
Total Internal Energy
22
23
Multiple Choice
In heat transfer, heat always flows from the _______________ substance to the _______________ substance.
warmer, cooler
cooler, warmer
24
Multiple Choice
As water in a freezer turns into ice, ___________.
the water absorbs energy from the air in the freezer.
the water absorbs the coldness from the air in the freezer.
the freezer air absorbs heat from the water.
the water neither absorbs nor releases energy
25
Multiple Choice
You take a cold can of soda off of a counter top. The counter top underneath the can feels colder than the rest of the counter. Which of the following explains this occurrence?
The cold has been transferred from the soda to the counter.
There is no heat energy left in the counter beneath the can.
Some heat has been transferred from the counter to the soda.
The heat beneath the can moves away into other parts of the countertop.
26
Multiple Choice
After cooking an egg in boiling water, you cool the egg by putting it into a bowl of cold water. Which of the following explains the egg’s cooling process?
Hot water moves from the egg to the water.
Cold water moves into the egg.
Energy is transferred from the water to the egg.
Energy is transferred from the egg to the water.
27
28
Fill in the Blanks

Type answer...
29
Fill in the Blanks
Type answer...
HEAT and TEMPERATURE
By Roberto Gonzalez
Show answer
Auto Play
Slide 1 / 29
SLIDE
Similar Resources on Wayground

19 questions
Scientific Notations
Presentation
•
9th Grade

21 questions
10s Photosynthesis lab KEY
Presentation
•
9th Grade

21 questions
Succession
Presentation
•
9th Grade

20 questions
Basic Chemistry
Presentation
•
9th Grade

23 questions
Photosynthesis & Cellular Respiration Review
Presentation
•
9th - 10th Grade

25 questions
Human Body Systems Pt 2
Presentation
•
9th Grade

21 questions
Cell Transport - Diffusion
Presentation
•
9th - 10th Grade

25 questions
Viruses
Presentation
•
9th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

10 questions
Exploring Chemical and Physical Changes
Interactive video
•
6th - 10th Grade